Translational Perioperative and Pain Medicine (ISSN: 2330-4871)
ARTICLE DOI: 10.31480/2330-4871/215
Research Article | Volume 12 | Issue 1 Open Access
Reframing the WHO Three-Step Analgesic Ladder: Embedding MT-Analgesia-3P Within the 4A5L-ALMT Multimodal Tuning Framework for Symptom-Cluster Management
Qin Yin1#, Fei-Fei Lu2#, Ming-Yue Cheng1, Yu-E Sun3, Jin-Feng Wang4* and Wei Cheng5*
1The Affiliated Hospital of Xuzhou Medical University, Xuzhou 221002, P.R. China
2The TCM Hospital Affiliated to Nanjing University of Chinese Medicine, Xuzhou 221009
3The Affiliated Hospital of Nantong University, No. 20 Xisi Road, Nantong, China
4The Suzhou Hospital of the Chinese Academy of Traditional Chinese Medicine - Xiyuan Hospital, Suzhou, China
5The Affiliated Huai'an No.1 People's Hospital of Nanjing Medical University, Huai'an 223300, P.R. China
#Contributed equally to this work
Wei Cheng, Huai'an No.1 People's Hospital of Nanjing Medical University, Huai'an 223300, China, Tel: +8618796205791, E-mail: 53974314@qq.com; Jin-Feng Wang, The Suzhou Hospital of the Chinese Academy of Traditional Chinese Medicine - Xiyuan Hospital, P.R. Suzhou, China, Tel: +8618168779112, E-mail: 709034013@qq.comEditor: Renyu Liu, MD; PhD; Professor, Department of Anesthesiology and Critical Care, Perelman School of Medicine at the University of Pennsylvania, Center of Penn Global Health Scholar, 336 John Morgan building, 3620 Hamilton Walk, Philadelphia, PA 19104, USA, Fax: 2153495078, E-mail: RenYu.Liu@pennmedicine.upenn.edu
Received: Nov 17, 2025 | Accepted: Jan 14, 2026 | Published: Jan 17, 2026
Citation: Yin Q, Lu FF, Cheng MY, et al. Reframing the WHO Three-Step Analgesic Ladder: Embedding MTAnalgesia-3P Within the 4A5L-ALMT Multimodal Tuning Framework for Symptom-Cluster Management. Transl Perioper Pain Med 2026; 12(1):846-859
Abstract
Background: The WHO three-step analgesic ladder offers a simple, linear escalation logic. In perioperative acute pain, chronic non-cancer pain (CNCP), and cancer pain/palliative care, however, escalation based solely on “pain intensity → drug intensity” often fails to balance functional recovery, sleep-mood-respiratory safety boundaries, and the risk of chronic postsurgical pain (CPSP).
Methods: Drawing on WHO/IASP/ERAS guidance and the broader evidence base, we propose 4A5L-ALMT as an analytic language to identify the system axes requiring “tuning” and the relevant decision layers. We then introduce MT-Analgesia-3P as an operational language-three graded implementation pathways (P1-P3)-to structure decisions, reassessment, and documentation.
Main content: Using three scenarios-acute postoperative pain, CNCP, and cancer pain/palliative care-we illustrate a pathway-based application centered on (1) triggers for entering P2/P3, (2) exit/de-escalation signals, and (3) a minimal indicator set for follow-up. An evidence-gradient approach distinguishes (1) established components (e.g., non-opioid multimodal analgesia, regional techniques, structured tapering, exercise/rehabilitation, CBT-I), (2) moderately supported elements used as optional contextual or risk information (e.g., symptom clusters, HRV), and (3) exploratory components that remain research-facing (e.g., digital phenotypes and dashboards).
Conclusions: MT-Analgesia-3P reframes the three-step ladder from a linear escalation scheme into a context-sensitive, three-pathway workflow designed to improve interdisciplinary communication and auditability for quality improvement. This framework is not a clinical guideline and does not replace existing guidance. It does not specify mandatory thresholds or titration algorithms. Prescribing within P2/P3-especially when opioids are involved-must follow local regulations and institutional governance, and must remain anchored to patient safety boundaries.
Keywords
WHO three-step analgesic ladder; Multimodal analgesia; Multimodal tuning; MT-Analgesia-3P; 4A5L-ALMT
Box 0 | Reader orientation (30 seconds)
Purpose: Provide a shared “grammar” for care teams-4A5L-ALMT as the analytic language plus MT-Analgesia-3P as the operational pathway language-supporting communication, follow-up documentation, and quality improvement.
Not a guideline: Does not replace WHO/IASP/ERAS or condition-specific guidelines; does not prescribe thresholds, dosing ceilings, titration algorithms, or a single mandatory escalation rule.
Evidence gradient: Prioritize established measures (non-opioid multimodal strategies, regional techniques, structured tapering, rehabilitation/exercise, CBT-I). Treat HRV and symptom clusters as optional context or risk signals. Treat digital phenotypes and dashboards as exploratory, research-oriented extensions.
Governance boundaries: Any opioid prescribing within P2/P3 must comply with laws, institutional policies, and monitoring capacity. Outside a palliative context, P3 does not imply “liberalized opioid escalation.”
Box 1 | Minimum-viable use (three questions)
Note: When resources are limited, start with the minimal indicator set. HRV, symptom-cluster tools, biomarkers, and similar measures should be treated as optional research enhancements-not requirements for routine care.
Q1-Set the primary axis (and the secondary axis): What is the dominant axis in this follow-up?
Pain processing; sleep-mood-cognition; autonomic-cardiovascular; inflammation-metabolism-posture/behavior-environment.
Q2-Choose the decision layer: Which layer is driving today’s decision?
L1 context and organ load; L2 mechanistic stratification; L3 symptom clusters; L4 indicator set; L5 intervention “knobs.”
Q3-Select a pathway and schedule reassessment:
Start with P1 as the foundation. Escalate to P2 only when needed and keep it time-limited (explicit goals + stop rules). If red flags emerge or the plan fails, move to P3 for higher-level evaluation and multidisciplinary management. De-escalate as soon as exit signals appear, and reassess on a defined cadence (e.g., 24-72 hours for acute settings or 2-4 weeks for longitudinal follow-up).
Introduction
Multimodal analgesia (MMA) has become increasingly systematized in perioperative medicine over the past two decades and, together with enhanced recovery after surgery (ERAS), has accelerated a clinical shift toward “less opioids, earlier mobilization, and faster recovery.” [1-4] Yet global surveys continue to show that inadequate postoperative pain control remains common. This is particularly evident after high-pain-burden procedures (e.g., spinal fusion and joint arthroplasty), where the downstream cascade-uncontrolled pain, disrupted sleep, worsening fatigue, limited activity, and rising complication risk-is often amplified. [5-7] Importantly, sleep is not a secondary outcome. It is a key amplifier of pain experience and a practical constraint that defines analgesic safety margins. For this reason, in the sections that follow we summarize sleep-pain-respiratory considerations strictly in terms of their decision implications, retaining only information that directly informs P1-P3 escalation, de-escalation, and reassessment.
The WHO three-step analgesic ladder remains valuable because it is intuitive, easy to teach, and scalable in resource-limited settings. Its linear escalation, however, is largely framed around “pain intensity → medication intensity,” with less explicit attention to time course, mechanism-based stratification, symptom clusters, and safety boundaries-variables that are central to contemporary analgesia and recovery medicine. We therefore propose a Multimodal Tuning (MT) perspective: analgesia is treated as a dynamic, cross-system, multi-objective process-balancing pain, sleep, function, and safety-rather than a one-way increase in drug potency.
To reduce communication burden and make follow-up documentation auditable, we introduce two complementary “languages” (Figure 1).
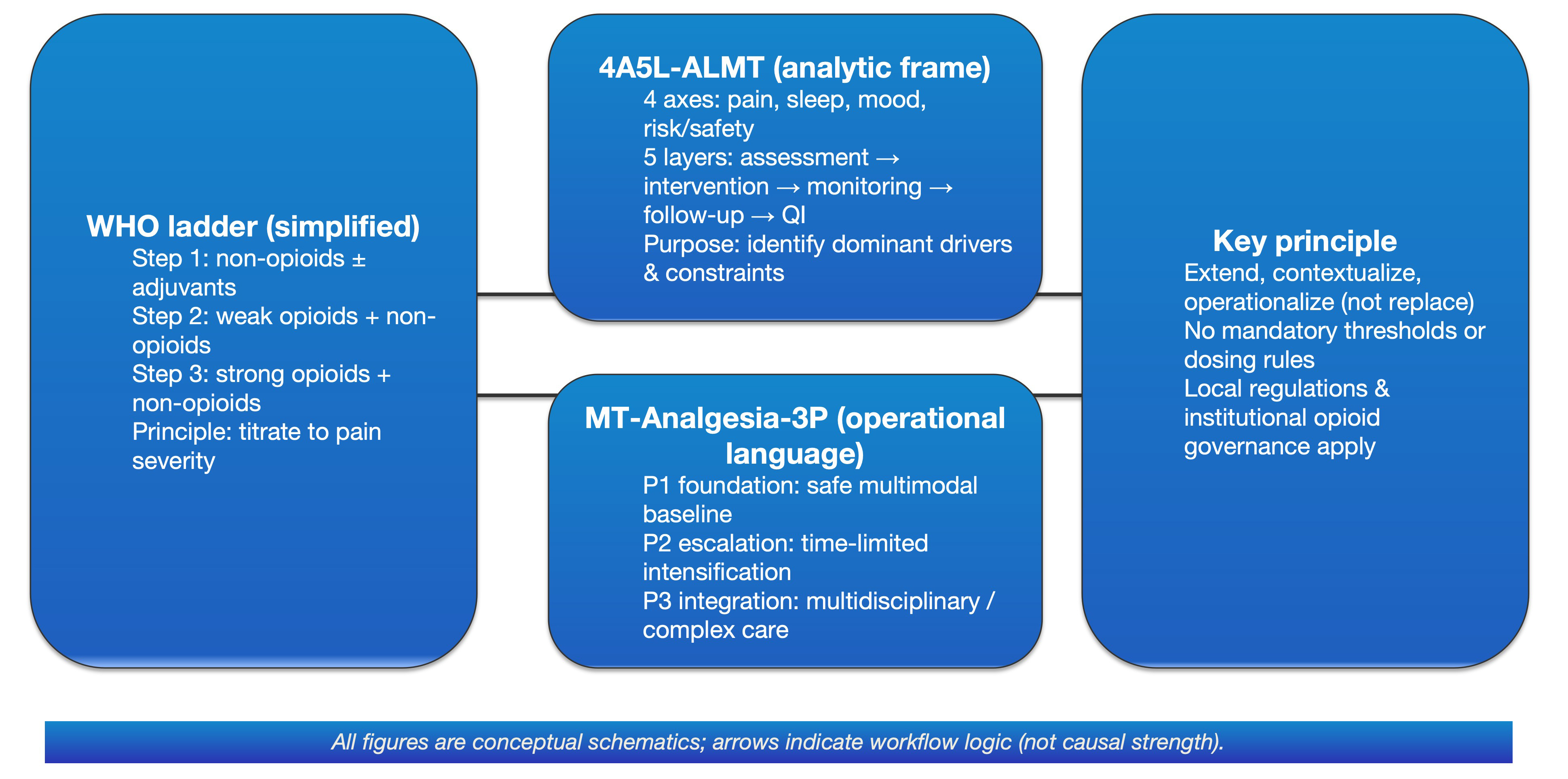
Figure 1: Conceptual map: embedding the WHO analgesic ladder into 4A5L-ALMT (analysis framework) and MT-Analgesia-3P (operational language).
The WHO ladder is used as a familiar reference point, but the intent is to extend, contextualize, and operationalize—not replace—existing guidance. The 4A5L-ALMT layer helps identify primary and secondary axes and match decisions to the strength of evidence; MT-Analgesia-3P translates this assessment into a documentable P1–P3 workflow with planned follow-up and reassessment. Arrows indicate the logical sequence of documentation and review, not dose ceilings, titration rules, or threshold-based algorithms. Final clinical choices should follow disease-specific guidelines, local regulations, and institutional governance.
Abbreviations and full terms
WHO:World Health Organization
4A5L-ALMT: Four Axes × Five Layers–Analytic Language for Multiscale Tuning
4A:Four Axes
5L:Five Layers
MT-Analgesia-3P:Multimodal Tuning Analgesia–Three Pathways
MT:Multimodal Tuning
P1 / P2 / P3:Pathway 1 / Pathway 2 / Pathway 3
QI:Quality Improvement
4A5L-ALMT (Four Axes × Five Layers-Analytic Language for Multimodal Tuning) serves as the analytic language. At any time point, it structures analgesia-relevant information into: the system axes that require parallel tuning (4A) and the decision layer where the primary problem resides (5L). The output is a shared, reviewable statement that can be checked and reproduced across clinicians and visits (Table 2).
Table 2: 4A5L-ALMT × MT-Analgesia-3P: minimum operational essentials.
|
Component |
Clinical question to answer |
Minimum information (low burden) |
Typical output/action (example) |
|
4A (four axes) |
Which system axes require tuning? |
Pain-processing axis: nociceptive / neuropathic / central sensitization [8-11]; sleep-emotion-cognition axis: insomnia/OSA/COMISA and stress-related factors [12-18,21-23]; autonomic-cardiovascular axis: HRV/nocturnal rhythms (optional risk signal) [19,20]; perioperative environment/process factors (light/noise/night-time interruptions) and key sleep-management points [14,15]. |
Identify a “primary axis + secondary axis”; document one sentence in the clinical record using the “axis-layer-pathway” syntax (for communication and auditability). [2,4] |
|
5L (five layers) |
At which layer is the bottleneck? |
L1 organ load/etiology: procedure type, comorbidities, and risk governance [2,4]; L2 mechanisms: nociceptive/neuropathic/nociplastic pain [8-11] and stress-related mechanisms (optional background) [23]; L3 phenotypes: capture the “pain × sleep × mood/stress” combination rather than NRS alone [12-18,21,23]; L4 measures: NRS + brief sleep measure (ISI/short PSQI/single-item) + key safety signals [4-6,12-18]; L5 interventions: non-opioid + regional techniques + sleep/behavioral support as the base; opioids as a time-limited tuning knob [1-6,14,15]. |
Assign tasks to responsible roles (anesthesia/surgery/nursing/rehab/psychology); prioritize the most likely 1-2 layers first to avoid “doing everything without focus”. [2,4] |
|
3P (three pathways) |
How intense should implementation be right now? |
P1 baseline: non-opioid agents + regional/local techniques + rehabilitation and basic sleep management [1-4,14,15]; P2 tuning: time-limited opioids/adjuvants on top of P1, with parallel monitoring of sleep-breathing safety and adverse events (especially in OSA/COMISA) [4-6,16-18]; P3 boundary & integration: when pain trajectory deviates / safety margin narrows / complications are suspected, trigger review and multidisciplinary integration rather than simply escalating doses [8-11,16-20]. |
Define entry/exit conditions; document escalation/de-escalation rationale and composite endpoints (pain × sleep × function × safety). [2-4,12-18] |
|
Time window |
Which time window are we in? |
Acute (≤72 h) / subacute (2-8 weeks) / chronic (≥3 months); focus on the acute-to-chronic transition risk and the CPSP concept [8-9]. |
Build follow-up time points into the pathway (post-op 24 h/72 h; 2/4/8 weeks; 3-6 months) and record the minimum indicator set at each node. [2-4,8,9] |
|
Composite endpoints |
Which outcomes are we trying to improve? |
Suggested minimum composite endpoint: pain intensity (including movement-evoked pain) + sleep quality/recovery + function/activity + key safety signals (sedation/respiration/delirium, etc.). [4-7,12-20] |
At each time point, anchor escalation/de-escalation to 2-3 priority indicators (e.g., NRS + brief sleep score + first ambulation/training milestone). [2-4,12-18] |
|
Evidence gradient |
Which elements are required vs optional? |
Three tiers: well-established (guidelines/consensus) [3,4]; sleep-pain bidirectionality and COMISA (moderate to strong) [12-18]; HRV/nocturnal rhythms as optional risk signals (evolving) [19,20]; stress-related mechanisms (explanatory background; avoid causal claims) [23]. |
Explicitly label exploratory elements as a research agenda rather than mandatory requirements to avoid being interpreted as an algorithm/decision tree. [3,4,19,20] |
|
Quality improvement (QI) |
How do we know the framework helps? |
Prioritize accessible KPIs: opioid use, analgesia target attainment, time to first ambulation, brief sleep score, adverse events; iterate using PDCA within ERAS/pathways. [2,4] |
Pilot small → collect feedback → iterate; produce a low-cost one-page template and a QI dashboard (not a decision algorithm). [2,4] |
Note: This table provides example “minimum operational essentials”. Citations support the evidence context for each component; medication choices and monitoring requirements should continue to follow local guidelines, regulations, and institutional governance.
Box 1. Reader orientation
4A5L: structure information (primary axis/layer/metrics) to generate an auditable record.
3P: choose implementation intensity (P1 baseline—P2 time-limited tuning—P3 boundary integration) and document entry/exit criteria and reassessment cadence.
How to use: the clinical team answers “What is the primary axis? Which layer is the bottleneck? Which pathway should we take?” to start; the rest (digital phenotypes/dashboards/QI) can be added stepwise as conditions allow.
MT-Analgesia-3P (Three Pathways for Multimodal Tuning Analgesia) serves as the operational language. It answers a practical question: at a given stage, what implementation intensity is appropriate for allocating analgesic resources? Practice intensity is organized into three switchable pathways. P1 (foundation pathway) emphasizes non-opioid multimodal strategies, regional techniques, rehabilitation, and basic sleep management as the base. P2 (tuning pathway) permits time-limited, reversible intensification-opioids and adjuvants are explicitly goal-driven, with defined stop rules, and entry/exit decisions are guided by parallel feedback across pain, sleep, function, and safety. P3 (boundary and integration pathway) is triggered when complications are suspected, techniques fail, or safety margins narrow; it prioritizes reassessment and integrated management, with the aim of achieving comfort and function within safety boundaries rather than simply increasing dose. We reiterate that this framework is intended to extend, contextualize, and operationalize existing ladders and guidelines-not to replace them. Its purpose is to provide a consistent documentation language to support teamwork and quality improvement.
Throughout this manuscript, “tuning” is used strictly as an organizing metaphor for information, reassessment, and governance. It is not an algorithm that generates prescriptions. All specific therapeutic decisions must still follow disease-specific guidelines, prescribing information, and institutional policy.
Methodologically, this is a narrative, framework-oriented review. We did not conduct exhaustive PRISMA-style searching or quantitative synthesis, and we do not quantify heterogeneity; accordingly, selection and interpretive bias are possible. To mitigate this risk, we prioritize higher-level evidence (guidelines, systematic reviews, large RCTs) and explicitly label evidence gradients where controversies exist [3,4]. Prior concepts from the author group are used as communication tools rather than as evidentiary conclusions, and we encourage independent external appraisal. Elements that lack sufficient validation (e.g., multi-axis digital phenotypes, wearables, and dashboard-based follow-up) are consistently labeled as “emerging/exploratory” and framed as a research agenda-not a clinical requirement. When autonomic markers such as HRV are mentioned, they are offered only as optional contextual or risk information for selected high-risk patients; they are not presented as routine monitoring requirements, and their use must be tailored to the clinical condition, medications, monitoring capacity, and local governance.
Across the acute-to-chronic continuum, time is the central dimension that requires the ladder to be re-contextualized. Chronic postsurgical pain (CPSP) is recognized in ICD-11 as a distinct diagnostic entity, and sustained nociceptive input can drive central sensitization and pain amplification [8-11]. Lowering NRS at a few time points, therefore, is not equivalent to reducing risk of persistence. In patients with pre-existing chronic pain, sleep disturbance, or elevated psychosocial risk, planning should shift earlier-into the preoperative period-and evolve into an iterative “postoperative tuning plan” over time [4,12]. Sleep and pain are bidirectionally coupled, [12-15] and comorbid insomnia and obstructive sleep apnea (COMISA) signals a narrower respiratory safety margin and heightened opioid-related risk [16-18]. Accordingly, this framework treats sleep/respiratory risk, safety margin, and mood/cognitive context as boundary conditions for P2/P3 decisions. The intent is to reduce imbalances created by one-dimensional medication escalation and to ensure that follow-up and quality improvement can be recorded and audited in a consistent, reviewable way.
Method statement and limitations (narrative framework review)
Readers should treat this manuscript as a structured template for communication, follow-up documentation, and governance. Disease-specific management must remain anchored to current guidelines, prescribing information, and institutional policy.
For this reason, we did not perform PRISMA-based exhaustive retrieval or quantitative pooling of all available studies. Evidence selection may be influenced by publication bias, the dispersion of relevant literature across disciplines, and author experience. To reduce bias, we present content using an “evidence-gradient + minimal falsifiable predictions” approach: we separate measures that are broadly implementable now from elements that should be treated only as risk signals or research-facing extensions (Table 1), and we repeatedly state the boundaries of the framework in both the main text and figure legends (non-threshold, non-algorithmic, non-prescriptive).
Table 1: Distribution of evidence gradients across the 4A system axes and the P1-P3 pathways (example; to prevent over-inference).
|
Dimension / component |
Well-established (mature) |
Moderate support |
Emerging / exploratory (research agenda) |
|
Pain-processing axis (core to P1/P2) |
Non-opioid foundations; regional / fascial plane blocks; multimodal combinations [1-4] |
Symptom clusters as an outcome-assessment framework [4,12,21,22] |
Automated decision support based on multi-axis data [12,19,20] |
|
Sleep-emotion-cognition axis |
CBT-I / sleep hygiene; psychological support; exercise prescription and rehabilitation [2-4,12,16-18,21,22] |
Brief scales / trajectories for follow-up stratification [12,15,18,21,22] |
App- or wearable-enabled continuous follow-up and individualized recommendations |
|
Autonomic-cardiovascular axis (safety boundary) |
Routine vital signs; sedation and respiratory monitoring (per guidelines) [3,4] |
HRV and nocturnal heart-rate rhythms: optional risk signaling [19,20] |
Novel autonomic biomarkers: require prospective validation [19,20,23] |
|
Inflammation-metabolism-posture/environment axis |
Exercise and weight management; environmental optimization; ERAS elements [2-4,23] |
Inflammatory / metabolic markers to support stratification when clinically indicated [23] |
End-to-end multi-axis digital phenotyping and dashboarded QI closed-loop systems [2,4] |
Note: This table is an example distribution to emphasize evidence gradients and appropriate boundaries of use; specific adoption and monitoring requirements should follow local guidelines and governance.
1) Established: non-opioid and regional analgesia as the foundation, with structured de-escalation and rehabilitation/psychological interventions as appropriate (CBT-I when available).
2) Moderate/emerging: symptom clusters and selected autonomic/digital follow-up tools are mainly used for risk signaling and research validation and should be implemented cautiously in line with local guidance and governance conditions.
This is a narrative and conceptual framework review. Its goal is to propose an operational “language-phenotype-pathway-reassessment” structure that translates clinical decisions into an auditable shared vocabulary. It is not intended as a systematic review of intervention effects, nor as a guideline-level recommendation document.
A Simplified “Four Axes-Five Layers + Three Pathways” Structure: A/L Terminology and Overview
4A5L-ALMT is designed to make the problem explicit.
The “4A” are four system axes that often require parallel tuning: (1) pain processing; (2) sleep-mood-cognition; (3) autonomic-cardiovascular; and (4) inflammation-metabolism-posture/behavior-environment [1-4,12-15,19-23]. The “5L” are five decision layers: (L1) etiology/organ load and context; (L2) mechanistic stratification; (L3) symptom clusters/phenotype; (L4) indicator set; and (L5) intervention “knobs.” [3,4,8-11] Importantly, the output is not “which drug to add,” but an auditable statement-primary axis/secondary axis, the layer where the key problem resides, and the priority indicators-so teams share a common language for pathway selection and scheduled reassessment [2,4].
MT-Analgesia-3P is designed to close the loop. P1 establishes the foundation (core non-opioid multimodal care, regional techniques, and rehabilitation in the MMA/ERAS context). P2 is time-limited tuning that is explicitly reversible and stoppable, iterated under parallel feedback across pain × sleep × function × safety. P3 is triggered when safety margins narrow-because of respiratory risk, comorbidity burden, technical failure, or an atypical trajectory-and focuses on reassessment and integrated management within defined safety boundaries [1-4,12-20]. In brief, 4A5L answers “what should be assessed and at which layer is the problem,” while 3P answers “when, and at what implementation intensity.” [2-4,19-23] To reduce overreach, we also make explicit how mature, moderately supported, and exploratory elements distribute across the 4A axes and P1-P3 pathways (Table 1), separating measures ready for routine implementation from elements that should be treated only as optional risk signals or a research agenda.
The four system axes (4A)
The four axes can be summarized as follows:
- Pain-processing axis: peripheral nociceptive input, neuropathic mechanisms, and central sensitization [8-11].
- Sleep-mood-cognition axis: insomnia, COMISA, depression, anxiety, and catastrophizing [12,16-18,21,22].
- Autonomic-cardiovascular axis: HRV, nocturnal heart rate, blood pressure rhythms, and stress load [19,20,23].
- Inflammation-metabolism-posture/behavior-environment axis: obesity, systemic inflammation, sedentary behavior, postural dysfunction, ward noise/light exposure, and care-process factors [23].
The five information/Decision layers (5L)
The five layers can be framed as five questions:
L1-Etiology/organ load and context: procedure type, comorbid disease, and organ-function burden [1-4].
L2-Mechanisms: nociceptive, neuropathic, and nociplastic components, plus inflammation-metabolism-posture contributions [8-11,27-27].
L3-Phenotype/symptom clusters: for example, “postoperative pain + sleep loss + fatigue,” or “chronic low back pain + insomnia + low mood.” [28-32]
L4-Indicators: NRS; ISI/PSQI; PHQ-9/HADS; daily opioid dose; HRV; step count; BMI/inflammatory markers; and related measures [12,16-23,33-39].
L5-Interventions: how medications, nerve blocks, exercise/rehabilitation, psychological interventions, weight management, and environmental optimization can be combined as an actionable set of “knobs.” [1-4,21-27,35-39].
Three Cross-Cutting Implementation Dimensions (Follow-up and Quality Improvement)
For practical deployment, three horizontal dimensions help translate the framework into routine workflows:
Time staging: acute (e.g., first 72 postoperative hours), subacute (2/4/8 weeks), chronic (3-6 months), and annual follow-up [8-9].
Digital phenotyping: repeated measures across stages for pain, sleep, mood, function, and safety-optionally augmented by wearable-derived signals and core EHR variables [12-15,16-18,19,20].
Quality improvement: aggregating indicators at the ward/clinic level into a concise dashboard to compare different pathway choices and ladder-application strategies [2,4].
How MT-Analgesia-3P maps onto the four axes and five layers
Building on the “4A + 5L + implementation dimensions” structure, MT-Analgesia-3P organizes analgesic resource allocation into three graded pathways:
P1 (foundation pathway): anchored in etiology and mechanism assessment, P1 combines non-opioid medications, local/regional techniques, rehabilitation and posture-oriented interventions, and basic sleep/circadian support as the default base.
P2 (tuning pathway): layered on top of P1, opioids and analgesic adjuvants are treated as time-limited, reversible tuning knobs that can be withdrawn, with adjustments guided by a multidimensional coordinate system-pain, sleep, mood, function, and safety.
P3 (boundary and integration pathway): in high-pain-burden or complex settings, P3 integrates interventional analgesia, neuromodulation, optimization of routes of administration, and-when appropriate-deeper sedation/analgesia strategies within preset safety boundaries, using composite endpoints such as comfort, calmness, and ability to communicate as feedback signals [26,27,33-35,40-42].
To move P1-P3 from definitions to executable resource allocation and feedback loops, Figure 2 provides a conceptual MT-Analgesia-3P workflow (arrows represent logical sequencing, not causal strength).
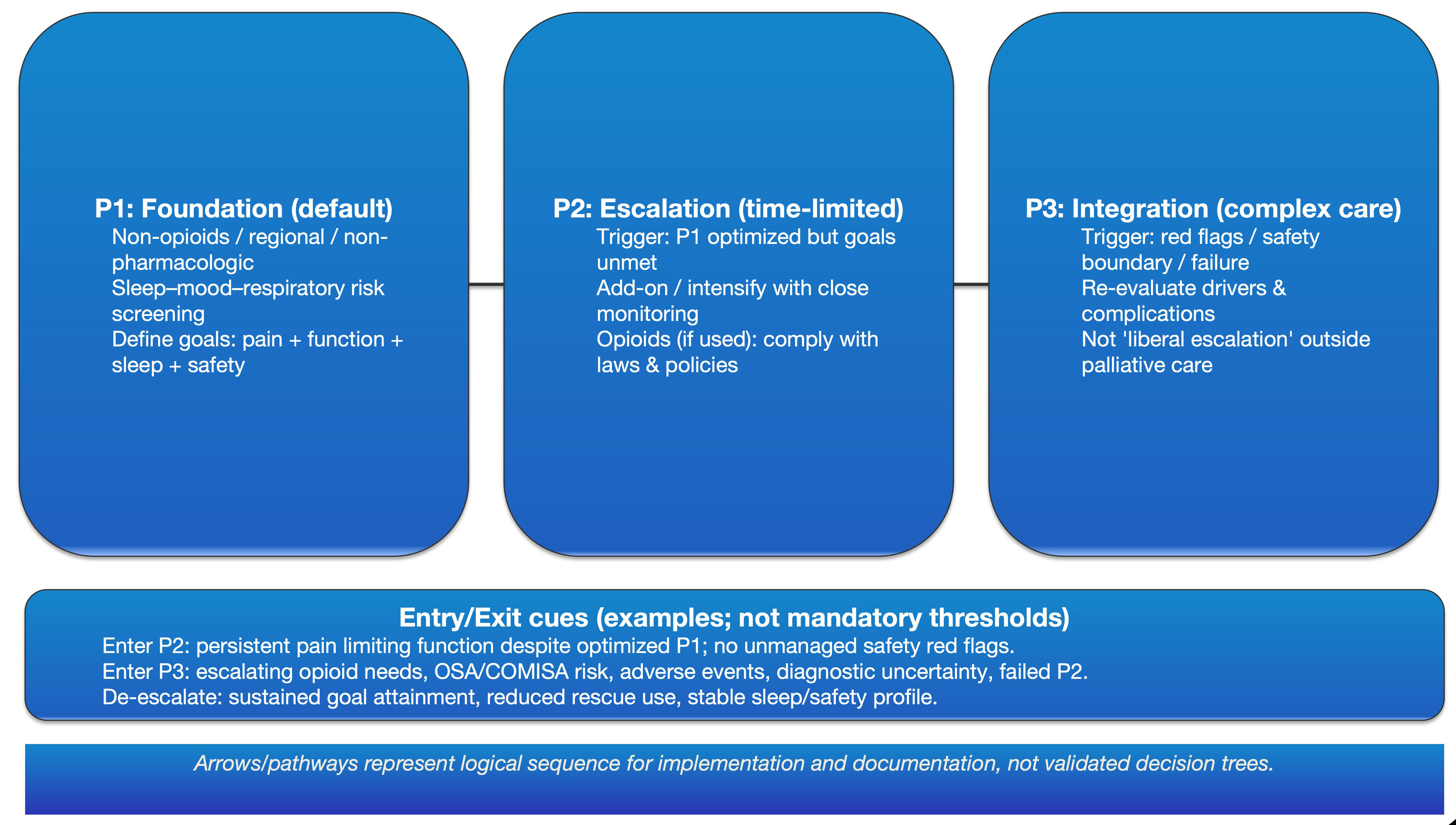
Figure 2: MT-Analgesia-3P workflow (conceptual; not an algorithm).
P1–P3 represent escalating levels of implementation intensity: P1 establishes a multimodal baseline; P2 adds time-limited, reversible intensification with explicit goals and stop rules; P3 triggers integrated management (e.g., complication screening, mechanism re-evaluation, multidisciplinary input, and governance). Arrows denote workflow and documentation order rather than causal strength, evidence weighting, dose limits, or titration algorithms.
Abbreviations and full terms
MT-Analgesia-3P:Multimodal Tuning Analgesia–Three Pathways
MT:Multimodal Tuning
P1 / P2 / P3:Pathway 1 / Pathway 2 / Pathway 3
OSA:Obstructive Sleep Apnea
COMISA:Comorbid Insomnia and Sleep Apnea
Where the WHO three-Step ladder sits within “4A + 5L + 3P”
Within this framework, the WHO ladder maps primarily to the pain-processing axis and the intervention layer (L5). Step 1 and parts of Step 2 (when combined with regional techniques) form key components of P1. Short-term opioids and selected adjuvants function mainly as P2 tuning knobs. Higher-dose opioids, complex administration routes, and interventional strategies are more often situated in P3, with their boundaries and feedback shaped by the other axes and layers [1-4,8-11,26,27,33-35,41]. In this view, “rewriting” the ladder across different scenarios reflects different implementation strategies within a shared multidimensional coordinate system, rather than mutually exclusive competing schemes.
Low-Resource Settings: Minimal vs Expanded Indicator Sets (Feasibility Statement)
Minimal indicator set (recommended across all resource levels): pain intensity and movement-evoked pain (NRS); function/mobility (e.g., mobilization milestones or a brief functional scale); perceived sleep restoration (single-item rating or short ISI/PSQI format); and opioid-related adverse effects plus sedation/respiratory risk (e.g., sedation score and a structured respiratory-risk screen) [4,12-15,16-18].
Expanded indicator set (optional when resources allow; useful for fine-tuning and research validation): HRV and nocturnal heart-rate rhythms, wearable-derived sleep parameters, inflammatory/metabolic biomarkers, symptom-cluster measures (fatigue, mood, catastrophizing), and digitized follow-up dashboards [19,20,28-32,36-39].
To avoid the misconception that this framework requires wearables or biomarkers, we provide low-resource-feasible examples of the minimal indicator set in the scenario maps (acute postoperative pain, CNCP, and cancer pain/palliative care). HRV, symptom-cluster instruments, inflammatory markers, dashboards, and decision-support tools are positioned strictly as optional research-facing enhancements-not as routine-care requirements (Figure 3).
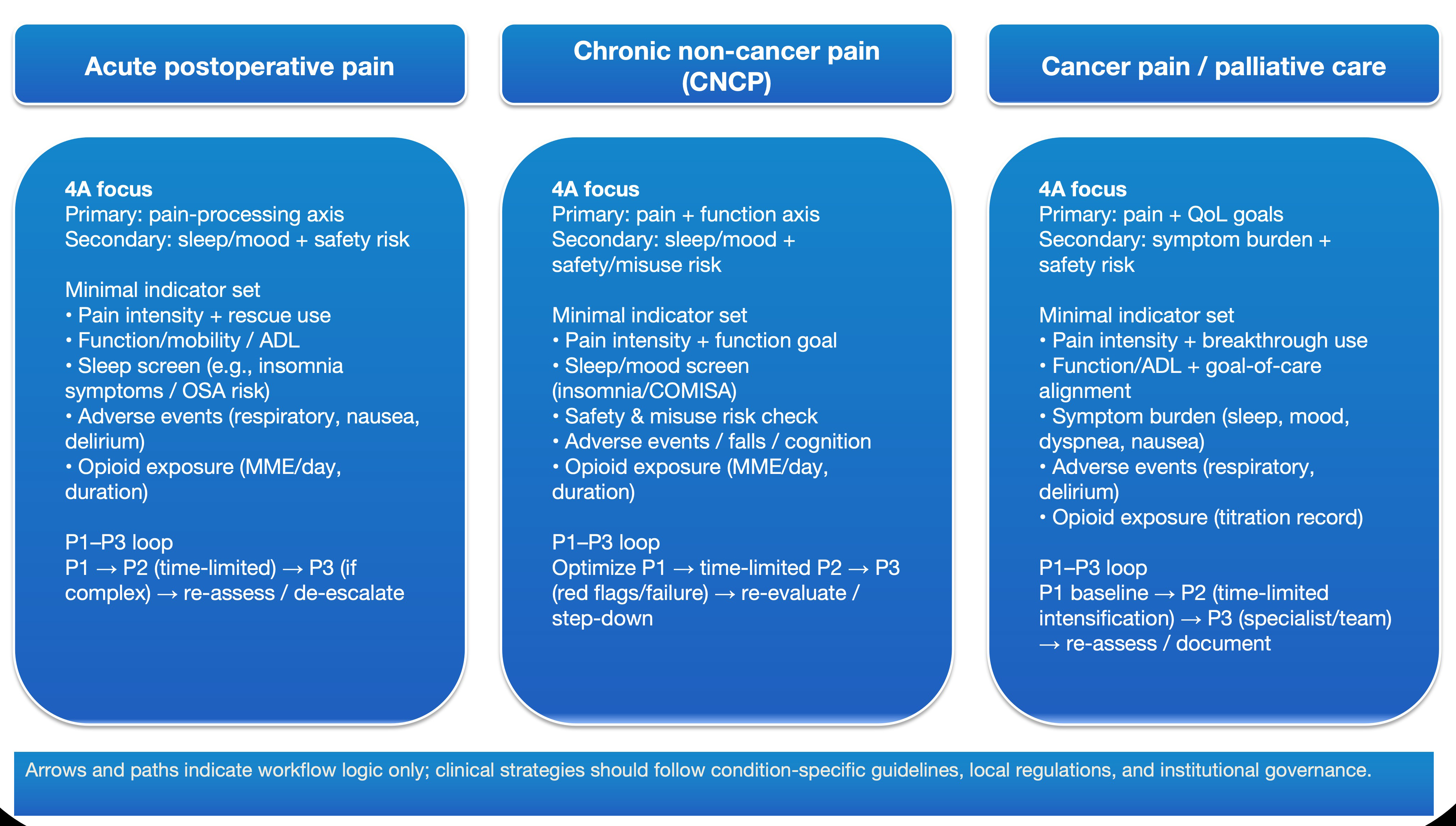
Figure 3: Scenario mapping across acute postoperative pain, CNCP, and cancer pain/palliative care (conceptual).
Across the three clinical scenarios, the 4A structure is used to define primary and secondary axes, select a feasible minimal indicator set, and run a P1–P3 escalation/de-escalation loop with follow-up. Arrows and pathways depict the intended logic for communication and tracking; specific therapeutic strategies remain anchored to disease-specific guidance, local regulations, and institutional governance.
Abbreviations and full terms
CNCP:Chronic Non-Cancer Pain
4A:Four Axes
ADL:Activities of Daily Living
OSA:Obstructive Sleep Apnea
MME:Morphine Milligram Equivalents
HRV:Heart Rate Variability
P1 / P2 / P3:Pathway 1 / Pathway 2 / Pathway 3
Symptom clusters and multidimensional Endpoints: What should the ladder aim for?
In oncology, the term “symptom cluster” describes a set of symptoms that co-occur over time and relate to one another-such as “pain-fatigue-depression” or “sleep disturbance-fatigue-low mood.” [28,29] Symptom clusters often explain quality-of-life differences better than any single symptom, and different clusters follow distinct trajectories and require different intervention strategies [30-32].
In spine surgery and chronic low back pain, systematic reviews indicate substantial heterogeneity in treatment effects. ERAS pathways can shorten length of stay and reduce complications, yet often pay less attention to “cluster endpoints” such as sleep, fatigue, mood, and long-term function [33,34]. Clinical guidelines for chronic low back pain consistently prioritize nonpharmacologic and non-opioid strategies, reserving opioids as a short-term option only after other approaches have failed [35].
Inflammatory and metabolic load matters as well. Across stages of nonspecific low back pain, inflammatory biomarker profiles can be complex, and epidemiologic evidence shows a robust association between obesity and chronic low back pain. Obesity may influence pain sensitivity and recovery through both systemic inflammation and mechanical loading [36-39].
For these reasons, the goal of the three-step ladder should not be reduced to a single target such as “NRS < 3.” Instead, it should be situated within a multidimensional space that includes pain, sleep, fatigue, mood, function, and the internal milieu (inflammatory/metabolic stress) [4,28-39]. At key time points (e.g., 72 hours postoperatively; 2/4/8 weeks; 3-6 months; or cancer follow-up milestones), the dominant symptom cluster can be summarized in one sentence, and each cluster can be anchored by two to three priority indicators (pain, sleep, function, calmness/comfort). Escalation and de-escalation should then be tied to these multidimensional anchors rather than to NRS alone [8,9,12-18,28-32].
Application Scenarios for MT-Analgesia-3P
Before moving into scenario-specific detail, we provide a practical “entry/exit” summary: triggers for escalation to P2/P3 and signals for de-escalation across acute postoperative pain, CNCP, and cancer pain/palliative care are consolidated in Table 3 (all framed as non-mandatory prompts, not threshold rules).
Table 3: Summary of prompts for entry to P2/P3 and exit/de-escalation across three scenarios (non-threshold; not an algorithm).
|
Scenario |
Enter P2 (triggers) |
Enter P3 (triggers) |
Exit/de-escalation (signals) |
|
Acute postoperative pain |
After P1 is implemented, clinically meaningful movement-evoked pain persists and impairs rehabilitation; or uncontrolled nocturnal pain clearly disrupts sleep after surgical/technical causes have been excluded. [1-7,12-15] |
Suspected complication or failure of analgesic technique; or escalating opioid needs with a narrowing safety margin (respiratory events, delirium, severe nausea/ileus, etc.); or a pain trajectory that is clearly off the expected course. [4,6,8-11,14-18] |
Functional goals are consistently met (ambulation/training achievable); nocturnal sleep and respiratory risk are controlled; opioid dose can be tapered as planned without rebound loss of control. [3-6,14-18] |
|
CNCP (chronic non-cancer pain) |
Limited to clearly defined short-term flares, and only if: (1) P1 optimization is complete; (2) a time limit and functional goals are pre-specified; (3) nonpharmacologic and non-opioid escalation is prioritized. [26-27,35-39] |
Existing moderate-to-high dose or long-term opioid exposure; or high-risk comorbidity (sleep/mood disorders, substance-use risk, etc.) / multi-mechanism phenotype requiring multidisciplinary re-review; or recurrent flares with inability to step down as planned. [21-23,26,27] |
Within each 3-6 month reassessment cycle: if functional benefit is stable, sleep/mood improves, opioid adverse effects are controlled, and taper conditions are met, initiate structured tapering and strategy switching. [21,22,26,27] |
|
Cancer pain / palliative care |
Moderate cancer pain or breakthrough pain continues to compromise sleep/activity or treatment adherence after P1, and maintaining wakeful communication is desired; enter P2 tuning under time-limited, reversible principles. [28-32,41] |
Severe cancer pain or advanced disease with complex symptom clusters/rapid progression; goals prioritize comfort and communicability, typically requiring coordinated oncology-pain-palliative care input (including route of administration and caregiver burden). [28-32,41] |
When adverse effects outweigh benefit, symptoms enter a relatively stable phase, or goals change, prioritize regimen simplification and reframe endpoints as “acceptable comfort and interactive capacity”. [41] |
Acute Postoperative Pain: From “Opioid-First” to “Regional Analgesia + Parallel Multi-Axis Tuning”
Escalate to P2 (triggers): P1 has been implemented, yet movement-evoked pain continues to block rehabilitation; or nocturnal pain remains uncontrolled and is clearly disrupting sleep after technical causes have been excluded.
Escalate to P3 (triggers): suspected complications or failure of the analgesic technique; rising opioid requirements accompanied by narrowing safety margins (respiratory events, delirium, severe nausea/ileus, etc.); or a pain trajectory that is clearly out of proportion to expectations.
Exit/de-escalate (signals): functional goals are consistently met (able to mobilize and train), nocturnal sleep and respiratory risk are acceptable, and opioids can be tapered as planned without rebound.
After high-pain-burden procedures such as spinal fusion and joint arthroplasty, a recognizable phenotype-“severe pain → insomnia/fragmented sleep → daytime fatigue”-is common [5-7,13]. In this framework, priorities shift as follows:
P1 (foundation): non-opioid multimodal therapy (e.g., NSAIDs/COX-2 inhibitors plus acetaminophen) combined with local/regional techniques (epidural, peripheral nerve blocks, fascial plane blocks), early rehabilitation, and basic sleep/circadian support. The aim is to achieve acceptable rest pain control and tolerable light-activity analgesia within the first 24-48 hours, while minimizing opioid dependence [1-4].
P2 (short-term tuning): once P1 is stable, patient-controlled analgesia (PCA) and short-acting opioids may be used briefly, with concurrent attention to three signal domains:
- nocturnal respiration and mental status;
- contextual stress/autonomic “background signals” (e.g., nocturnal heart rate/HRV when feasible);
- opioid-related adverse events (respiratory depression, delirium, etc.) [14,15,19,20,23].
As sleep and function improve, a taper plan should be documented immediately; extra caution is warranted in patients with OSA/COMISA or other respiratory vulnerability.[16-18]
P3 (boundary and integration): the core intent is not “dose liberation,” but a structured trigger for complication screening, mechanism reappraisal, and multidisciplinary integration. This includes reassessing for hematoma, infection, neurologic injury, hardware-related problems, and the integrity/coverage of the analgesic technique, before escalating interventions, routes of administration, or monitoring intensity [8-11]. Patients who require sustained high-dose opioids or repeated invasive interventions should be treated as high-risk for CPSP and enrolled into follow-up accordingly [8,9,26,27].
Operationally, the first 72 postoperative hours can be treated as a discrete “tuning unit.” Standardized documentation at 24/48/72 hours-pain, sleep, function, opioid dose, and key safety events-supports ward-level quality improvement (QI) comparisons across pathway choices [2-7,33,34].
Chronic Non-Cancer Pain (CNCP): Mechanism-Based Stratification and a “Reverse Ladder”
In CNCP, the key is not escalation, but an auditable de-escalation loop (Figure 4, the cross-scenario entry/exit card; CNCP is represented as a reverse-ladder logic).
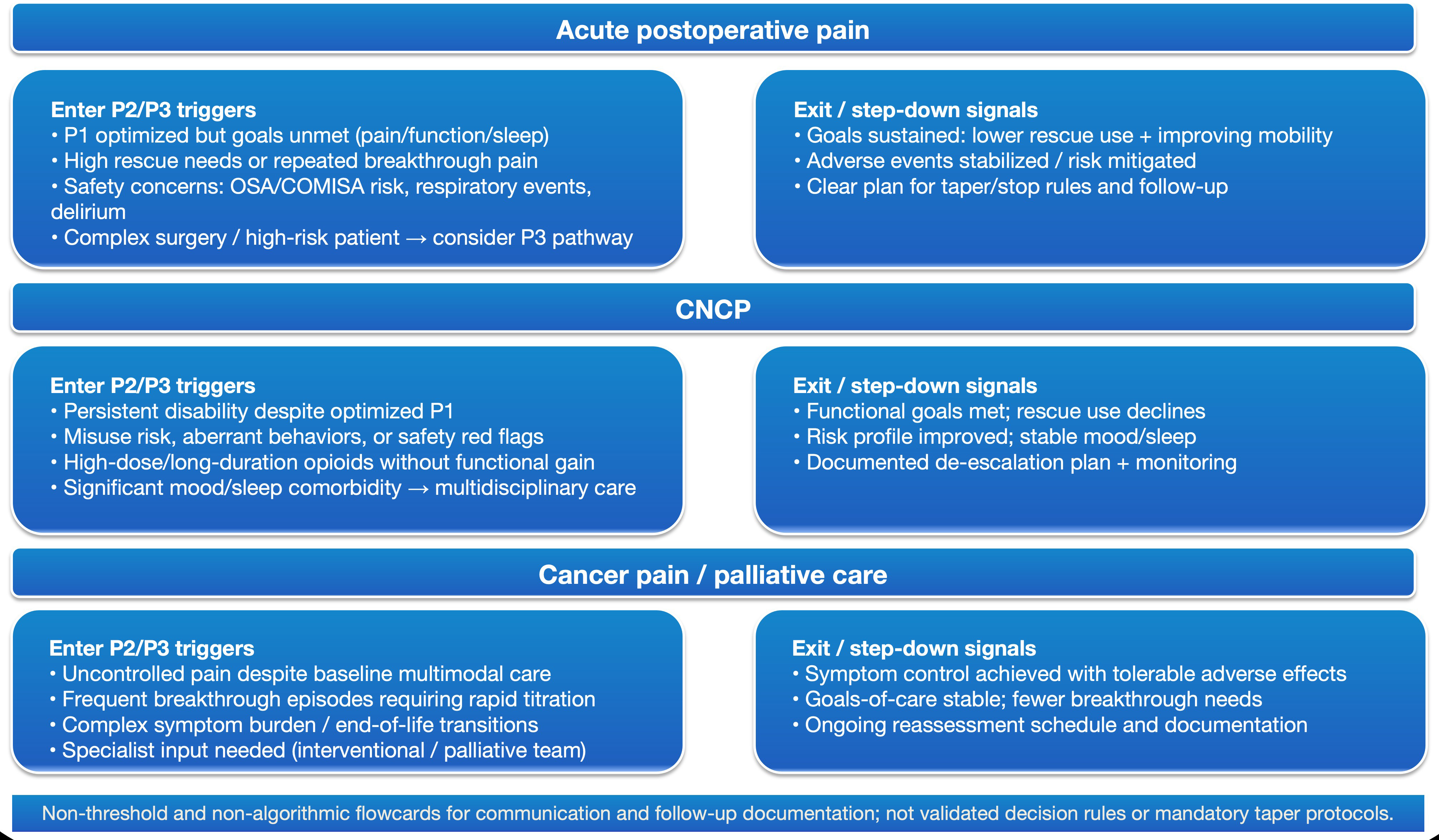
Figure 4: Entry triggers to P2/P3 and exit/de-escalation signals across three scenarios (conceptual).
This flowcard summarizes practical signals for when to initiate time-limited intensification (P2) or integrated multidisciplinary management (P3), and when it is appropriate to step down to baseline care (P1) after goals are achieved and risks are controlled. It is designed to support communication, follow-up documentation, and auditability—not to function as a threshold-based rule, dosing algorithm, or mandatory taper protocol.
Abbreviations and full terms
P2 / P3:Pathway 2 / Pathway 3
CNCP:Chronic Non-Cancer Pain
P1:Pathway 1
OSA:Obstructive Sleep Apnea
COMISA:Comorbid Insomnia and Sleep Apnea
Escalate to P2: restricted to short-term flares and only when (i) P1 has been optimized, (ii) a predefined time limit is set, and (iii) functional goals are explicit-preferably by stepping up nonpharmacologic and non-opioid strategies first [26,27,35].
Escalate to P3: established moderate-to-high dose or long-term opioid exposure; high-risk comorbidity burden or multi-mechanism phenotypes requiring multidisciplinary review; or recurrent flares that prevent planned step-down [21,22,26,27].
Exit/de-escalate: reassess every 3-6 months. When functional benefit is sustained, sleep/mood improve, and taper conditions are met, initiate structured tapering and strategy switching [26,27].
Mechanistically, a pragmatic stratification includes: nociceptive pain (opioids, if used at all, should be time-limited with an early taper point);[26,35] neuropathic pain (gabapentinoids and SNRI/TCA as primary options; opioids reserved for short rescue use);[40] and nociplastic/mixed mechanisms (prioritizing exercise, CBT-I, pain education, weight management, and behavioral interventions) [11,16,35-39]. Follow-up can run on a compact indicator set spanning pain × sleep × mood × function × safety, while clinic dashboards track taper success rates and functional gains over time [26,27].
Cancer Pain and Palliative Care: Comfort and Dignity as the Endpoint
Escalate to P2: moderate cancer pain or breakthrough pain that disrupts sleep or treatment adherence, when maintaining wakefulness and communication is important-using time-limited, reversible tuning [41].
Escalate to P3: severe cancer pain or rapidly evolving, complex symptom clusters in advanced disease, typically requiring coordinated input from oncology, pain, and palliative teams [41,42].
In this scenario, success is not defined solely by driving NRS toward zero, but by achieving comfort, calmness, and the ability to interact at an acceptable level of consciousness. Practicality of administration routes and care burden often matter more than fine dose optimization [41]. Data collection can be intentionally minimal-pain, perceived sleep restoration, fatigue/calmness, and key adverse events-with goals re-set dynamically as disease stage changes [41,42].
Implementation and Research Outlook: Making the Ladder “Work” Within a Multimodal Tuning Paradigm
How to Test “Whether It Works” in Real Practice
To keep the framework from remaining a conceptual diagram, we propose implementation-oriented evaluation across three interlocking levels (without introducing additional labels):
- At the individual prescribing level
- At the team and pathway level
- At the system and quality-improvement level
Across ERAS pathways, chronic pain clinics, and oncology/palliative teams, the starting point is a one-page template for high-frequency scenarios (e.g., spine/joint arthroplasty, chronic low back pain, cancer pain). The template should document the primary and secondary axes, the dominant symptom cluster, key time points, and the two to three priority indicators to track at each time point. Crucially, the reasons for stepping up or stepping down between P1-P3 should be written explicitly on the template rather than being buried implicitly in the medication list [2-4,26,27,33-35,41]. A ready-to-use example is provided in Table 4.
Multidisciplinary communication can be grounded using a small set of fixed questions: What are the primary and secondary axes? What time stage are we in? Do the key indicators support staying in P1, moving briefly into P2, or triggering P3 integration? [26,27,41,42] The goal is to make “axis-layer-pathway” the default sentence structure of daily rounds and case discussions, not something that appears only in figures.
Early implementation should prioritize low-burden metrics that can populate an auditable dashboard: perioperative opioid exposure, analgesia target attainment, time to first mobilization, and a simplified sleep rating. For CNCP, dashboards should emphasize opioid taper success and functional gain; for oncology/palliative care, calmness/comfort and unplanned utilization (ED visits/readmissions) are often more informative [2-7,26,2733-35,41,42]. When capacity allows, teams can progressively add HRV, wearable-based sleep/activity, and weight or inflammatory markers to build a multi-axis “digital phenotype” dataset for subsequent decision-support research [12,16-23,36-39].
In short, MT-Analgesia-3P is not “a new guideline that replaces guidelines.” It is a documentation and implementation language that helps existing evidence work more clearly within clinical collaboration, iterative reassessment, and QI loops: 4A/5L organizes the information, and P1-P3 allocates resources, repositioning the WHO ladder from a “dose staircase” into a multidimensional coordinate system spanning pain × sleep × function × safety [1-8,12,21-23,26,2733-35,41,42]. To minimize frontline burden, we provide a one-page template (Table 4) to support consistent recording and follow-up auditing.
Table 4: Clinician-friendly one-page implementation template (printable / embeddable in the medical record).
|
Field (Table 4) |
Content (citations retained) |
Suggested key references (for verification) |
|
Scenario (check one) |
□ Acute postoperative □ Chronic non-cancer pain (CNCP) □ Cancer pain/palliative care. [3,4,26,27,41,42] |
|
|
Goals (check and/or add) |
□ Analgesia □ Functional recovery □ Sleep/mood □ Reduce opioid-related risk □ Other: _____. [1-4,6,12-18,21,22,26,27,41,42] |
|
|
Minimum assessment (document/check) |
NRS / pain location and quality; function (walking/coughing/ADL); sedation/respiratory risk; prior opioid exposure; patient preferences/goals. [4-6,16-18,22,26,41] |
|
|
P1 key actions |
Prioritize non-opioid multimodal therapy (e.g., acetaminophen, NSAIDs/COX-2 inhibitors) + regional/local analgesic techniques; address symptom clusters (nausea, anxiety, insomnia, etc.) and basic sleep hygiene/circadian management in parallel. [1-4,12-18,21,22] |
|
|
P2 escalation triggers / exit conditions |
Trigger: after adequate P1, pain control remains inadequate or function is limited; use opioids/adjuvants as time-limited, reversible “tuning” with concurrent monitoring of respiratory/sedation risk and adverse events. Exit: stable improvement → taper/stop as planned to avoid long-term exposure. [4-6,16-18,26] |
|
|
P3 escalation triggers / exit conditions |
Trigger: red flags/suspected complication/very high risk/analgesic technique failure requiring individualized or multidisciplinary integration and mechanism re-evaluation; boundary: follow regulations and institutional policies (opioid governance/monitoring capacity); exit: risk decreases or reversible factors corrected → step down. [4,8,11,26,27,40-42] |
|
|
Follow-up and reassessment plan |
Reassessment time points: post-op 24-48 h / before discharge / 1-4 weeks (scenario-dependent); at each review, document key changes in “4A×5L” and monitor signals of acute-to-chronic transition risk. [2-4,8,9,14,15,26,27] |
|
|
QI documentation (auditable items) |
Document: reason for escalation; adverse events; taper plan; patient-reported outcomes (PROMs); readmission/return visits; costs/resource use (optional). [2-4,6,26,27,41,42] |
Evidence Gaps and Limits of Generalization: What to Prioritize Next
- Population specificity
Children and older/frail adults differ substantially in analgesic-sedative sensitivity, respiratory safety margins, and sleep architecture. Their minimal indicator sets, P2/P3 entry criteria, and tapering strategies require dedicated validation [12,16-18,26,27]. - Generalizability across health systems
While the framework language is intended to be culturally portable, implementation strategy and endpoint choice are shaped by resource density, prescribing governance, and access to rehabilitation. These factors require calibration through multicenter implementation-science studies [2,4]. - Long-term outcomes and tapering
High-quality prospective data that can be directly translated into outpatient workflows remain limited-particularly for transitions from postoperative prescribing to chronic use, long-term taper success in CNCP, and the relationship of tapering to composite endpoints that include sleep, mood, and function. We recommend composite outcomes such as “taper success + functional gain + safety events” and their inclusion in dashboards [21,22,26,27]. - Trade-offs versus simpler models
Platform models, ladder revisions, and reverse-ladder approaches can be advantageous for low-resource settings and rapid decisions. The added value of the present framework is an auditable “axis-layer-pathway” recording method for heterogeneous scenarios, enabling iterative refinement and quality control under uncertainty (Table 5) [33-36].
Table 5: Scenario-based comparison of ladder/platform/reverse-ladder models Vs MT-Analgesia-3P (for “gradient revision” trade-offs).
|
Approach/model |
Core decision logic (brief) |
Best-fit scenarios |
Strengths and limitations (this manuscript’s stance) |
|
WHO traditional three-step analgesic ladder (classic) |
Escalate pharmacotherapy by pain severity: non-opioids → weak opioids → strong opioids (with adjuvants/supportive measures as needed) [41] |
Cancer pain and resource-limited settings; when a fast, teachable prescribing logic is needed [41,42] |
Strength: simple and widely disseminated. [41] |
|
Simplified/revised ladder variants (common in the literature) |
Platform- or scenario-based two/three-step variants: emphasize early multimodal and regional techniques, reduce reliance on the “weak opioid” step, or adjust based on risk stratification. [1-4,14,15,26] |
Postoperative analgesia, ERAS, and opioid-risk-sensitive populations (e.g., OSA/COMISA, delirium/respiratory vulnerability). [1-4,5-7,14-18] |
Strength: reduces opioid exposure and aligns with ERAS and recovery goals. [1-4,5-7] |
|
De-escalation ladder (reverse ladder) |
Control pain and function first, then step down/taper based on benefit-risk balance with explicit reassessment and exit criteria. [26,27,35] |
CNCP, long-term opioid management, and populations at CPSP risk. [8,9,26,2735] |
Strength: naturally embeds tapering and reassessment loops; fits chronic disease management. [26,27,35] |
|
MT-Analgesia-3P (P1→P2→P3) embedded in 4A5L-ALMT |
P1: multimodal + regional-first; P2: short-term, controlled opioid/adjuvant tuning; P3: complexity/complications/multidisciplinary integration; 4A×5L documents “why escalate/when de-escalate”. [2-4,26,27,33-35,41] |
Applicable across all three scenarios (acute postoperative, CNCP, cancer pain/palliative care), especially for complex cases with “unclear safety boundaries + multidimensional goals”. [12-20,26-32,35,41,42] |
Strength: integrates risk-goals-follow-up-QI into a single language and reduces misuse; provides entry/exit prompts and a minimal indicator set. [2-4,12-18,26,27] |
Falsifiable hypotheses and a minimal validation roadmap
The contribution of this work is not a new guideline, but a clearer separation between analytic language (4A5L) and operational language (3P), scenario-specific entry/exit prompts, and explicit inclusion of evidence gradients and low-resource minimal sets to reduce misuse risk.[2-4,26,27,35] To support testable implementation research, we propose the following optional, falsifiable hypotheses with minimal design thresholds:[2,4]
- Effectiveness
- Safety and opioid governance
- Cost-effectiveness
- Population specificity
- Digital validation
Adding 3P prompts and a one-page template to standard ERAS reduces CPSP incidence or improves a composite endpoint (pain × function × sleep).
Design: pragmatic RCT or stepped-wedge cluster trial [2-4,8,9,12-18].
The approach reduces discharge opioid intensity and/or persistent use without increasing acute loss of control or readmissions.
Endpoints: MME, persistent use rate, respiratory adverse events, unscheduled revisits.[4-6,16-18,26]
Across systems with different resource density, the “minimal indicator set + one-page template” captures most of the benefit.
Evaluation: cost-utility (QALYs) or cost-effectiveness (reduced readmissions/ED utilization) [2-4,35].
Pediatric and older/frail populations require distinct entry/exit thresholds and safety boundaries; stratified analyses or dedicated cohorts can optimize indicator sets and safety margins [16-18,26].
Using an app/dashboard as a recording and feedback tool (not a prescribing algorithm) improves follow-up adherence and completion of QI loops.
Pathway: feasibility pilot first, followed by effectiveness testing [2-4,12,19,20].
To reduce bias from cultural and system-level differences, we recommend prospective multicountry or multi-region implementation studies using a shared “axis-layer-pathway” data dictionary, with prescribing governance, rehabilitation access, and pain-expression norms treated as explanatory variables in analytic models [2,4].
Statement and dissemination guidance (Avoiding Misreading as a “Guideline/Algorithm”)
In submission materials, the “minimal indicator set + P1-P3 closed loop” can be positioned as the core low-resource-feasible value proposition. Wearables, biomarkers, and decision-support tools should be explicitly labeled as research or QI extensions to prevent reviewers from interpreting them as routine requirements.
For clinical roll-out or teaching, three boundary statements should be presented together:
- this framework supports communication and follow-up documentation and does not replace guidelines;
- Figure 3, Figure 4 are non-threshold, non-algorithmic workflow cards and do not constitute prescribing rules or mandatory taper protocols;
- any opioid, sedation, or neuromodulation strategy must comply with local regulations and institutional governance, with appropriate risk disclosure and monitoring.
Conclusion
This manuscript reinterprets the WHO three-step analgesic ladder through a Multimodal Tuning (MT) lens. 4A5L-ALMT serves as an analytic language that frames the primary problem and a minimal indicator set at any time point using “system axes (4A) × decision layers (5L).” MT-Analgesia-3P serves as an operational language that organizes analgesia, sleep, function, and safety within a single feedback loop through graded implementation pathways (P1-P3). Using scenario-specific entry/exit prompts for acute postoperative pain, CNCP, and cancer pain/palliative care, the framework aims to reduce multidisciplinary communication burden, improve the auditability of follow-up and QI, and support actionable “less opioids, better recovery” practice within regulatory and patient-safety boundaries. Its effectiveness, safety, and cost-effectiveness still require confirmation through implementation-science studies across different populations and health systems.
Funding:
National Natural Science Foundation of China (Project Approval Number: 81200858); Jiangsu Province 333 High-level Talent Training Project [Certificate No.: (2022) No. 3-10-007].
Clinical Trials from Nanjing Drum Tower Hospital, Affiliated Hospital of Medical School, Nanjing University, the Huai'an Matching Assistance Special Project (2024-2025).
Conflict of interest: None.
AI assisted declaration: The authors used generative AI tools (e.g., ChatGPT, OpenAI) to assist in language polishing and formatting. All intellectual content, analyses, and conclusions are solely those of the authors.”
Presentation: None Declared.
References
- Kehlet H, Dahl JB. The value of “multimodal” or “balanced analgesia” in postoperative pain treatment. Anesth Analg. 1993;77(5):1048–1056. doi:10.1213/00000539-199311000-00030.
- Kehlet H, Wilmore DW. Evidence-based surgical care and the evolution of fast-track surgery. Ann Surg. 2008;248(2):189–198. doi:10.1097/SLA.0b013e31817f2c1a.
- McEvoy MD, Scott MJ, Gordon DB, et al. American Society for Enhanced Recovery (ASER) and Perioperative Quality Initiative (POQI) joint consensus statement on optimal analgesia within an enhanced recovery pathway for colorectal surgery: part 1—from the preoperative period to PACU. Perioper Med (Lond). 2017;6:8. doi:10.1186/s13741-017-0064-5.
- Chou R, Gordon DB, de Leon-Casasola OA, et al. Management of postoperative pain: a clinical practice guideline. J Pain. 2016;17(2):131–157. doi:10.1016/j.jpain.2015.12.008.
- Apfelbaum JL, Chen C, Mehta SS, Gan TJ. Postoperative pain experience: results from a national survey suggest postoperative pain continues to be undermanaged. Anesth Analg. 2003;97(2):534–540.
- Gan TJ. Poorly controlled postoperative pain: prevalence, consequences, and prevention. J Pain Res. 2017;10:2287–2298. doi:10.2147/JPR.S144066.
- Kunze KN, Movasagghi K, Rossi DM, et al. Systematic review of sleep quality before and after arthroscopic rotator cuff repair: are improvements experienced and maintained? Orthop J Sports Med. 2020;8(12):2325967120969224. doi:10.1177/2325967120969224.
- Schug SA, Lavand’homme P, Barke A, et al. The IASP classification of chronic pain for ICD-11: chronic postsurgical or posttraumatic pain. Pain. 2019;160(1):45–52. doi:10.1097/j.pain.0000000000001413.
- Voscopoulos C, Lema M. When does acute pain become chronic? Br J Anaesth. 2010;105(Suppl 1):i69–i85. doi:10.1093/bja/aeq323.
- Woolf CJ. Central sensitization: implications for the diagnosis and treatment of pain. Pain. 2011;152(3 Suppl):S2–S15. doi:10.1016/j.pain.2010.09.030.
- Kosek E, Cohen M, Baron R, et al. Do we need a third mechanistic descriptor for chronic pain states? The term “nociplastic pain”. Pain. 2016;157(7):1382–1386. doi:10.1097/j.pain.0000000000000506.
- Finan PH, Goodin BR, Smith MT. The association of sleep and pain: an update and a path forward. J Pain. 2013;14(12):1539–1552. doi:10.1016/j.jpain.2013.08.007.
- Fielden JM, Gander PH, Horne JG, et al. An assessment of sleep disturbance in patients before and after total hip arthroplasty. J Arthroplasty. 2003;18(3):371–376. doi:10.1054/arth.2003.50056.
- Rampes S, Ma K, Divecha Y, et al. Postoperative sleep disorders and their potential impacts on surgical outcomes. J Biomed Res. 2019;34(4):271–280. doi:10.7555/JBR.33.20190054.
- Yuan X, Ju Z, Zhang X, Yin X. Perioperative sleep disturbance in surgical patients: a concept analysis. Clin Nurs Res. 2024;33(6):493–501. doi:10.1177/10547738241241905.
- Sweetman A, Lack L, McEvoy RD, et al. Co-morbid insomnia and sleep apnea (COMISA): prevalence, consequences, and treatment. Brain Sci. 2019;9(12):371. doi:10.3390/brainsci9120371.
- Sweetman A, Melaku YA, Clark J, et al. Prevalence and associations of co-morbid insomnia and obstructive sleep apnoea in the general population. Sleep Med. 2021;82:9–17. doi:10.1016/j.sleep.2021.03.023.
- Luyster FS, Buysse DJ, Strollo PJ Jr. Comorbid insomnia and obstructive sleep apnea: challenges for clinical practice and research. J Clin Sleep Med. 2010;6(2):196–204. doi:10.5664/jcsm.27772.
- Koenig J, Jarczok MN, Ellis RJ, et al. Heart rate variability and experimentally induced pain in healthy adults: a systematic review. Eur J Pain. 2014;18(3):301–314. doi:10.1002/j.1532-2149.2013.00357.x.
- Zamunér AR, Barbic F, Knebel F, et al. Relationship between sympathetic activity and pain intensity in fibromyalgia. Clin Exp Rheumatol. 2015;33(1 Suppl 88):S53–S57.
- Meints SM, Edwards RR. Evaluating psychosocial contributions to chronic pain outcomes. Prog Neuropsychopharmacol Biol Psychiatry. 2018;87(Pt B):168–182.
- Hooten WM. Chronic pain and mental health disorders: shared neural mechanisms, epidemiology, and treatment. Mayo Clin Proc. 2016;91(7):955–970. doi:[
- McEwen BS, Kalia M. The role of corticosteroids and stress in chronic pain conditions. Metabolism. 2010;59(Suppl 1):S9–S15. doi:10.1016/j.metabol.2010.07.012.
- Bushnell MC, Čeko M, Low LA. Cognitive and emotional control of pain and its disruption in chronic pain. Nat Rev Neurosci. 2013;14(7):502–511. doi:10.1038/nrn3516.
- Tracey I, Mantyh PW. The cerebral signature for pain perception and its modulation. Neuron. 2007;55(3):377–391. doi:10.1016/j.neuron.2007.07.012.
- Turk DC, Wilson HD, Cahana A. Treatment of chronic non-cancer pain. Lancet. 2011;377(9784):2226–2235. doi:10.1016/S0140-6736(11)60402-9.
- Gatchel RJ, McGeary DD, McGeary CA, et al. Interdisciplinary chronic pain management: past, present, and future. Am Psychol. 2014;69(2):119–130. doi:10.1037/a0035514.
- Miaskowski C, Barsevick A, Berger A, et al. Advancing symptom science through symptom cluster research: expert panel proceedings and recommendations. J Natl Cancer Inst. 2017;109(4):djw253. doi:10.1093/jnci/djw253.
- Kirkova J, Aktas A, Walsh D, Davis MP. Cancer symptom clusters: clinical and research methodology. J Palliat Med. 2011;14(10):1149–1166. doi:10.1089/jpm.2010.0507.
- Baden M, Lu L, Drummond FJ, et al. Pain, fatigue and depression symptom cluster in survivors of prostate cancer. Support Care Cancer. 2020;28(10):4813–4824. doi:10.1007/s00520-019-05268-0.
- Palesh O, Aldridge-Gerry A, Zeitzer JM, et al. Sleep disruption in breast cancer patients and survivors. J Natl Compr Canc Netw. 2013;11(12):1523–1530. doi:10.6004/jnccn.2013.0170.
- Rodgers C, Hooke MC, Ward J, Linder LA. Symptom clusters in children and adolescents with cancer. Semin Oncol Nurs. 2016;32(4):394–404. doi:10.1016/j.soncn.2016.08.006.
- Xu W, Ran B, Luo W, Li Z, Gu R. Is lumbar fusion necessary for chronic low back pain associated with degenerative disk disease? A meta-analysis. World Neurosurg. 2021;146:298–306. doi:10.1016/j.wneu.2020.11.121.
- Elsarrag M, Soldozy S, Patel P, et al. Enhanced recovery after spine surgery: a systematic review. Neurosurg Focus. 2019;46(4):E3. doi:10.3171/2019.1.FOCUS18700.
- Qaseem A, Wilt TJ, McLean RM, et al. Noninvasive treatments for acute, subacute, and chronic low back pain: a clinical practice guideline from the American College of Physicians. Ann Intern Med. 2017;166(7):514–530. doi:10.7326/M16-2367.
- Morris P, Ali K, Merritt M, et al. A systematic review of the role of inflammatory biomarkers in acute, subacute and chronic non-specific low back pain. BMC Musculoskelet Disord. 2020;21(1):142. doi:10.1186/s12891-020-3154-3.
- Shiri R, Karppinen J, Leino-Arjas P, et al. The association between obesity and low back pain: a meta-analysis. Am J Epidemiol. 2010;171(2):135–154. doi:10.1093/aje/kwp356.
- Okifuji A, Hare BD. The association between chronic pain and obesity. J Pain Res. 2015;8:399–408. doi:10.2147/JPR.S55598.
- Galassi T, Pacheco-Barrios K, Gianlorenco AC, Fregni F. Obesity and systemic inflammation disrupt the compensatory role of physical activity in chronic pain conditions. Biomedicines. 2025;13(5):1111. doi:10.3390/biomedicines13051111.
- Dworkin RH, O’Connor AB, Backonja M, et al. Pharmacologic management of neuropathic pain: evidence-based recommendations. Pain. 2007;132(3):237–251. doi:10.1016/j.pain.2007.08.033.
- Caraceni A, Shkodra M. Cancer pain assessment and classification. Cancers (Basel). 2019;11(4):510. doi:10.3390/cancers11040510.
- Temel JS, Greer JA, Muzikansky A, et al. Early palliative care for patients with metastatic non–small-cell lung cancer. N Engl J Med. 2010;363(8):733–742. doi:10.1056/NEJMoa1000678.
Table of Contents
- Abstract
- Keywords
- Introduction
- Methods Overview: A Minimal, Auditable Strategy for a Narrative Review
- The Sleep Anesthesia Coma Continuum and Phenotypes of Unconsciousness
- Integrating Consciousness Theories with Anesthetic States: Testable Predictions and Contrasts
- The Systems Network Layer: The NAEI Network from Axes to Outcomes
- Evolution of Perioperative Medicine and the Role of the Anesthesiologist
- Nonpharmacologic Neuromodulation: VNS and Electroacupuncture as Tuning Tools
- The Micro Level: Yin Yang and Quantum Concepts as a Hypothesis Generating Layer
- Cellular/Molecular Anesthesiology, Personalization, and Neurotoxicity: Limits of the Evidence and a Research Agenda
- Monitoring and Closed-Loop Control: From EEG to Connectivity and NAEI Trajectories
- The Three-Layer Multiscale Tuning Model and Working Definition
- Limitations and Future Directions
- Conclusion
- Funding
- Conflicts of Interest
- Funding
- AI Use Statement
- Figure 1
- Figure 2
- Figure 3
- Figure 4
- Table
- Table
- Table
- Table
- Table
- References