Translational Perioperative and Pain Medicine (ISSN: 2330-4871)
ARTICLE DOI: 10.31480/2330-4871/214
Research Article | Volume 12 | Issue 1 Open Access
Anesthesia As a Multiscale Tuning Process: From Clinical State Control to the NAEI Network and Yin Yang/Quantum Heuristics
Qin Yin1#, Fei-Fei Lu2#, Ming-Yue Cheng1, Yu-E Sun3, Jin-Feng Wang4* and Wei Cheng5*
1The Affiliated Hospital of Xuzhou Medical University, Xuzhou 221002, P.R. China
2The TCM Hospital Affiliated to Nanjing University of Chinese Medicine, Xuzhou 221009
3The Affiliated Hospital of Nantong University, No. 20 Xisi Road, Nantong, China
4The Suzhou Hospital of the Chinese Academy of Traditional Chinese Medicine - Xiyuan Hospital, Suzhou, China
5The Affiliated Huai'an No.1 People's Hospital of Nanjing Medical University, Huai'an 223300, P.R. China
#Contributed equally to this work
Wei Cheng, Huai'an No.1 People's Hospital of Nanjing Medical University, Huai'an 223300, China, Tel: +8618796205791, E-mail: 53974314@qq.com; Jin-Feng Wang, The Suzhou Hospital of the Chinese Academy of Traditional Chinese Medicine - Xiyuan Hospital, P.R. Suzhou, China, Tel: +8618168779112, E-mail: 709034013@qq.comEditor: Renyu Liu, MD; PhD; Professor, Department of Anesthesiology and Critical Care, Perelman School of Medicine at the University of Pennsylvania, Center of Penn Global Health Scholar, 336 John Morgan building, 3620 Hamilton Walk, Philadelphia, PA 19104, USA, Fax: 2153495078, E-mail: RenYu.Liu@pennmedicine.upenn.edu
Received: Nov 17, 2025 | Accepted: Jan 14, 2026 | Published: Jan 17, 2026
Citation: Yin Q, Lu FF, Cheng MY, et al. Anesthesia As a Multiscale Tuning Process: From Clinical State Control to the NAEI Network and Yin Yang/Quantum Heuristics. Transl Perioper Pain Med 2026; 12(1):831-845
Abstract
Background: General anesthesia is often defined as a drug-induced, reversible state of unconsciousness with analgesia and suppression of reflexes. This single-layer description does not capture anesthesia-related, time-structured effects across scales, including brain network organization, autonomic balance, endocrine stress signaling, immune-inflammatory responses, and recovery-relevant outcomes.
Objective: Within a perioperative precision-medicine perspective, we integrate network-neuroscience evidence with a nervous-autonomic-endocrine-immune (NAEI) systems framework and propose a three-layer working definition that reframes anesthesia as a reversible multiscale tuning process. An optional Yin–Yang/quantum discussion is included strictly as a cross-disciplinary heuristic to generate falsifiable, proxy-anchored research questions, not as mechanistic proof or a basis for clinical recommendations.
Methods: Conceptual narrative review (2000-2025). We did not follow PRISMA procedures or perform meta-analysis. To improve auditability, we used an evidence-gradient approach with multi-database searching plus citation tracking and constrained key claims to measurable anchors (EEG, HRV, inflammatory/endocrine markers, and behavioral/clinical outcomes).
Results: The proposed model couples three layers: (1) a clinical-operational layer of controllable “knobs” for state control; (2) a systems-network layer describing NAEI trajectories across perioperative time windows; and (3) a micro-level hypothesis-generating layer that remains strictly conditional and non-causal.
Conclusions: An NAEI-guided, three-layer tuning paradigm supports explicit knob-readout-outcome mapping, phenotype-network-outcome modeling, and hypothesis-driven study designs for endpoints such as perioperative neurocognitive disorders, infection/inflammatory burden, and recovery trajectories.
Keywords
General anesthesia, Consciousness, Brain networks, Neuro-autonomic-endocrine-immune (NAEI) networks, Perioperative outcomes
Introduction
Classic reviews define general anesthesia as a drug-induced, reversible depression of the central nervous system. Clinically, it presents as loss of consciousness, analgesia, attenuated reflexes, and muscle relaxation [1-3]. With advances in systems neuroscience and functional neuroimaging, anesthesia has been placed within a “sleep-anesthesia-coma continuum,” alongside natural sleep and pathological coma as distinct brain states [1-3]. In this view, unresponsiveness does not necessarily equal unconsciousness. Behavioral non-response must be separated from changes in subjective experience and awareness [4]. Anesthesia is therefore not only a clinical tool. It also functions as an in-vivo model for studying consciousness. By tracking how awareness is lost and returns under controlled drug exposure, competing theories can be linked to measurable proxies and mapped onto clinically observable state transitions [5,6].
Over the past two decades, many studies have examined how anesthetics reshape large-scale brain networks. Across agents, general anesthesia can weaken information integration between hub regions and alter the topology of the default mode and fronto-parietal networks, yielding a broadly consistent “network disconnection” pattern [7-9]. During early propofol-induced loss of consciousness, networks can fragment rapidly and long-range connections can collapse, whereas recovery often coincides with reconnection [10]. High-density EEG has further defined reproducible spectral and phase features during loss and return of consciousness, providing objective signal endpoints for state transitions [11]. Together, these findings suggest that a single “receptor inhibition” narrative is not sufficient; a more useful account organizes multiscale readouts into measurable, reviewable loops that can support cross-study comparison and hypothesis testing [8,9,11].
Against this background, we describe anesthesia as a multiscale tuning process. It includes controllable clinical “knobs,” such as anesthetic depth, analgesia-sedation balance, ventilation and circulation targets, sleep/circadian support, and neuromodulation parameters. It also includes systems-level readouts, such as EEG connectivity and complexity, HRV and autonomic reflexes, inflammatory and hormonal profiles, and behavioral and patient-reported outcomes (PROs) [11-13]. At the circuit and cellular level, anesthesia and sleep share core substrates, including the ascending arousal system, the thalamic reticular nucleus, and cortico-thalamic loops. Animal studies suggest that GABAergic neurons in the thalamic reticular nucleus participate in propofol- and isoflurane-related state control [12-14]. At the synaptic and microcircuit level, intravenous and inhaled agents modulate GABAergic and glutamatergic transmission, ion channels, and excitation-inhibition balance, thereby altering effective information propagation across networks [15,16].
To avoid relying on behavior alone, anesthesia research has also developed behavior-independent metrics. Examples include TMS-EEG-based perturbational complexity measures (e.g., PCI), interpretable deep-learning approaches that separate arousal from content-related EEG components, and integrated-information-related estimates derived from high-density recordings [17-19]. During recovery, network efficiency and posterior alpha patterns have been used to characterize return trajectories. Resting-state studies under ketamine show selective disruption within the default mode and fronto-parietal networks, supporting network-level phenotyping across anesthetic states [20,21]. Major theories of consciousness-including IIT, GNW, and recurrent processing-provide testable, competing predictions about which signals should change with consciousness and how those changes relate to behavior [22-25]. Work on dynamic complexity and on dissociating “environment awareness” from “self-related awareness” further suggests that content, access, and report can diverge in anesthesia settings [26-30].
This paper is a concept-integrative framework/position piece for perioperative clinicians and researchers. The primary evidentiary weight is placed on the clinical-operational layer and the NAEI systems-network layer, expressed through an audit-ready “knobs-readouts-outcomes” language. An optional micro-level Yin-Yang/quantum discussion (Section 8) is included solely to generate falsifiable questions linked to measurable proxies; it is not presented as mechanistic proof, a causal model, or a basis for clinical recommendations, and it may be skipped without loss of the clinical-NAEI framework.
Methods Overview: A Minimal, Auditable Strategy for a Narrative Review
This article is a conceptual narrative review and theoretical synthesis. Given the cross-scale scope of the topic, we did not conduct a PRISMA-guided systematic review or quantitative meta-analysis. To reduce selective-citation risk and improve auditability, we adopted a minimal, auditable strategy built on three principles:
Multi database, cross disciplinary coverage
We performed parallel searches and citation tracking across four framework-aligned blocks: (i) anesthesia-consciousness-brain networks (EEG/fMRI connectivity, complexity, PCI); (ii) NAEI physiology (HRV/autonomic, inflammatory and endocrine readouts); (iii) perioperative outcomes across time windows (PND/delirium, infection, recovery trajectories, and selected longer-term endpoints including cancer outcomes); and (iv) controversial micro-level proposals (microtubules/Orch-OR, quantum biology, oxygen-pathway), treated strictly as hypothesis-generating [7-13,17-21,26-28].
Evidence gradient labeling
Framework elements were stratified as strong, moderate, or speculative, and correlational findings were not framed as causal. When discussing consciousness theories, we organized arguments as competing predictions linked to measurable proxies [22-27-29]. Briefly, we label evidence as strong when supported by convergent findings and/or quantitative synthesis, moderate when supported by consistent but context-dependent clinical/mechanistic literature, and speculative when primarily theoretical, model-based, or limited to indirect/animal evidence.
Priority of measurable anchors and falsifiability
All cross-layer claims were tied to at least one measurable anchor (EEG, HRV, inflammatory/endocrine markers, behavioral or PRO outcomes), enabling verification or falsification. For anesthetic brain states, readouts were anchored to established EEG state markers and monitoring evidence, avoiding reliance on a single scalar index [11,28-35]. In contested domains, we applied a causal-guarding rule: mechanistic verbs were avoided unless supported by strong evidence; other wise we used conditional language and paired each claim with at least one measurable proxy and an explicit falsification route.
Minimal search trace
We searched PubMed/MEDLINE, Embase, Web of Science Core Collection, and Scopus (2000-2025), supplemented by backward/forward citation tracking; the last search was performed on 26 December 2025. Example (database-adapted) strings included: (“general anesthesia” AND consciousness AND (EEG OR connectivity OR complexity OR PCI)); (anesthesia AND HRV); (anesthesia AND (IL-6 OR CRP OR cortisol)); and (anesthesia AND cancer AND (recurrence OR survival)). We prioritized consensus statements, major reviews, and landmark studies that link claims to measurable anchors; contentious areas were presented with competing interpretations and non-causal language. Where our prior work is cited, it serves only to document terminology/concept provenance; major claims are supported primarily by independent external evidence. Examples of evidence-gradient labeling and their corresponding measurable anchors are summarized in (Table 1).
Table 1: Evidence gradient: Multi-scale framework elements and measurable anchors.
|
Evidence level |
Representative elements (how used in this article) |
Representative measurable anchors |
Falsifiable / testable pathways (examples) |
|
Strong evidence |
Anesthesia-related EEG spectra/burst suppression, connectivity and network fragmentation; reproducible signal markers of loss and recovery of consciousness; perioperative trajectories of HRV and inflammatory/stress responses [10,11,20,31,32,34,35] |
EEG spectra/complexity, BIS/entropy; fMRI functional connectivity; HRV (rMSSD, HF, LF/HF); inflammatory markers such as IL-6 and CRP [11,31-33] |
Compare network readouts across drugs/techniques under matched depth strata; use time-window trajectories to predict recovery quality/delirium risk [11,20,21,33,34] |
|
Moderate support |
Multi-axis NAEI coupling framework; directional effects of neuromodulation (VNS/taVNS, electroacupuncture/TEAS) on autonomic and inflammatory readouts (context-dependent) [11,20,31,32,35] |
HRV + inflammation/cytokines + stress hormones; sleep continuity; QoR, pain, delirium [11,20,31,32,33] |
Prospective cohort or embedded randomization: intervention → NAEI trajectories → outcomes; mediation/path analysis (not equivalent to causal proof) [26,31,32,33] |
|
Speculative (hypothesis-generation layer) |
Orch-OR, quantum coherence, microtubule oscillations; “Yin-Yang” as a heuristic mapped to measurable physiological variables [22-25,28-30] |
EEG complexity/perturbational response, HRV-inflammation coupling, energy-metabolism markers [17-19,31,32] |
Formulate competing predictions in “if…then…” form; compare theories on perturbation-response and complexity-drop patterns; requires independent empirical validation [17,22-25] |
Finally, the micro-level Yin-Yang/quantum content (Section 8) is intentionally optional and speculative: it is included solely to generate falsifiable questions linked to measurable proxies and is not presented as mechanistic proof or a basis for clinical recommendations.
The Sleep Anesthesia Coma Continuum and Phenotypes of Unconsciousness
Within the sleep-anesthesia-coma continuum, propofol-induced loss of consciousness is commonly associated with rapid fragmentation of functional networks and collapse of long-range connectivity, whereas recovery is typically accompanied by network re-integration [9,10]. High-density EEG captures reproducible spectral and phase features during both loss and return of consciousness, providing objective endpoints for state transitions [10,11].
Mechanistically, anesthesia and sleep share core substrates, including the ascending arousal system, the thalamic reticular nucleus, and cortico-thalamic circuits. GABAergic neurons in the anterior thalamic reticular nucleus have been implicated in propofol- and isoflurane-related state control [12-14]. Across agents, intravenous and inhalational anesthetics modulate GABAergic and glutamatergic transmission and voltage-gated channels, shifting excitation-inhibition balance and weakening effective information transfer [15,16]. These multiscale findings reinforce a key distinction: unresponsiveness does not necessarily equal unconsciousness, and internal experience may persist during behaviorally unresponsive states [4,15].
Accordingly, behavior-independent readouts have been developed. Perturbational complexity (PCI) uses TMS-EEG to quantify post-stimulus spatiotemporal complexity as a proxy for consciousness capacity, while interpretable deep-learning approaches and integrated-information-related metrics (φ) further separate arousal from content-related components across states [17-19]. During recovery, network efficiency and posterior alpha activity track return trajectories. Resting-state fMRI under ketamine shows selective disruption within default mode and fronto-parietal networks. Cross-species evidence further suggests that general anesthesia reduces the “uniqueness” of functional connectivity, shifting the brain toward lower-diversity configurations [8,10,20,21]. Together, these measures provide a network-level toolkit for characterizing anesthetic brain states and recovery trajectories, and they motivate the theory-driven, falsifiable contrasts summarized next (Table 2).
Table 2: Evidence boundaries, core disputes, and testable proxies for major consciousness theories under anesthesia (e.g., IIT, GNW, and related accounts).
|
Theory / framework |
Core prediction (simplified) |
Measurable readouts under anesthesia (examples) |
Testable control designs (examples) |
|
IIT (Integrated Information Theory) |
Unconsciousness primarily reflects reduced information integration; decreases in complexity/integration should track loss of consciousness [22,17] |
PCI; complexity/entropy metrics; integrated functional connectivity; perturbation-response spatiotemporal complexity [17-19] |
Compare complexity-drop patterns across agents and depth strata; at matched behavioral unresponsiveness, test whether complexity differs [17,23] |
|
GNW (Global Neuronal Workspace) |
Consciousness depends on ignition of a globally accessible workspace; unconsciousness reflects failed global broadcasting/blocked ignition [25,22] |
Frontoparietal long-range connectivity; ignition-like EEG/ERP signatures; task-evoked global broadcast signals (paradigm-dependent) [25,23] |
Under task paradigms, compare loss and recovery of ignition/broadcast markers; designs that dissociate these markers from overt report ability [25,29] |
|
Recurrent processing / posterior-cortex-dominant view (example) |
Conscious content relies more on recurrent processing in posterior cortex; frontal cortex is more related to reporting [23,29] |
Posterior-cortex connectivity and local–global interactions; report vs no-report paradigm differences [23,29] |
No-report controls: test links between posterior readouts and experiential cues without explicit report [23,29] |
Integrating Consciousness Theories with Anesthetic States: Testable Predictions and Contrasts
Major theories of consciousness (e.g., IIT, GNW, recurrent processing) differ in structural assumptions and dynamics and therefore generate distinct predictions about which signals should change under anesthesia, the magnitude of those changes, and whether neural markers can dissociate from behavior [22,25]. A central NCC debate concerns whether experiential content depends primarily on posterior cortices, while frontal regions are more strongly linked to report, access, and executive control [23,29]. GNW emphasizes ignition and maintenance of a globally accessible workspace, prioritizing long-range connectivity and ignition-like dynamics as key readouts [22-25].
To avoid unresolved theory debate, we translate disagreements into operational contrasts. Evidence boundaries, disputed points, and measurable proxies are presented side by side, with anesthesia-specific comparison paths outlined in Table 2. This format enables re-evaluation, external validation, and falsification within harmonized time windows and acquisition frameworks [22-25]. To illustrate the approach, we provide an anesthesia-specific contrastive testing framework using IIT and GNW as examples (Table 3).
Table 3: Testable predictions in anesthesia: IIT vs GNW.
|
Theory |
Core claim (ultra-brief) |
Key prediction for anesthesia |
Priority measurable anchors (examples) |
Falsifiable prediction (examples) |
Main limitations / caveats |
|
IIT (Integrated Information Theory) |
Consciousness corresponds to the amount/structure of integrated information [22,17] |
Anesthesia markedly reduces integration/complexity; perturbation-evoked global integration decreases [17-19] |
PCI/perturbational response; EEG complexity and long-range coupling; spread of evoked potentials [17-19] |
If behavior is unresponsive under anesthesia but PCI remains near awake levels, one must explain “unresponsiveness ≠ unconsciousness” or model mismatch [4,15,17] |
Metrics depend on paradigms and SNR; different agents may produce distinct complexity trajectories [11,18] |
|
GNW (Global Neuronal Workspace) |
Consciousness depends on global broadcasting/ignition in the workspace [25,22] |
Anesthesia suppresses global broadcasting/ignition (e.g., frontoparietal); local processing may persist while global access is limited [25,23] |
Frontoparietal functional connectivity; P3/ignition-related ERP; cross-regional phase synchrony/effective connectivity [25,23] |
If global connectivity is strongly suppressed yet stable ignition-like activity and report-like cues persist, GNW predictions require revision or stratification [25,29] |
Connectivity measures are vulnerable to motion/EMG artifacts; different agents may “disconnect” different pathways [11,8] |
|
Consensus layer (this article) |
Organize hypotheses around measurable anchors rather than adjudicating theories [22,24] |
Translate disputes into controlled experiments and data acquisition plans [22-25] |
EEG (depth + complexity) × behavioral responsiveness; add HRV/inflammation as NAEI coupling readouts when needed [11,17,31,32] |
If predictions are systematically inconsistent with multicenter data, update the model or delimit its domain [22,24] |
This article does not deliver a final verdict; it provides a framework for asking falsifiable questions [22,24] |
A key limitation follows: no single brain readout can discriminate among theories. Consequently, theory testing in anesthesia is most informative when coupled to perioperative system-level anchors (e.g., HRV, inflammatory and stress axes) and to outcome-relevant time windows, enabling an auditable multiscale validation chain and NAEI-based modeling of recovery variability [11,20,31,32,35]. This coupling provides the bridge to the systems-network layer developed in the next section.
The Systems Network Layer: The NAEI Network from Axes to Outcomes
HRV, inflammation, and the NAEI axes
To capture anesthesia’s broad effects on brain-body systems, a purely central nervous system view is insufficient. The nervous, autonomic, endocrine, and immune-inflammatory axes must be integrated as the NAEI network [1,35]. HRV, as a dynamic marker of sympatho-vagal balance, has been widely reviewed in anesthesia assessment, hemodynamic risk stratification, and postoperative recovery [31,35].
Perioperative inflammation is shaped jointly by anesthetic drugs and techniques, surgical stress, pain, and sleep disruption, and should be interpreted within defined time windows and covariate frameworks rather than attributed to single factors. Meta-analyses suggest anesthetic approaches may be associated with differences in IL-6 and CRP, but effects are context dependent [32,35]. In oncologic surgery, the “critical window” hypothesis makes pathways from anesthetic/analgesic strategy to immune-inflammatory modulation and longer-term outcomes testable, but requires multi-axis covariate control and re-auditable endpoints [33,34,36-38].
Accordingly, we treat inflammation and immunity not as conclusions but as measurable anchors within the NAEI network that connect stress load, analgesic quality, sleep continuity, and infection or recovery trajectories. Operationalization follows a “term → node → proxy → falsifiable criterion” mapping (Table 4). In practice, dominant modules (N, A, E, or I) and priority anchors can be identified within each perioperative time window, guiding knob selection and reassessment. Figure 1 provides a minimal schematic of key readouts, controllable knobs, and outcome endpoints across the N/A/E/I modules, embedding Table 4 within a single auditable loop.
Table 4: NAEI network: Modules, key measurable anchors, and controllable tuning knobs.
|
Heuristic term |
NAEI node mapping |
Measurable proxy variables (examples) |
Falsifiable criteria (examples) |
|
“Yang-predominant / high arousal” |
N/A (brain network + autonomic) |
Higher EEG complexity or increased β/γ activity; lower HRV-HF with higher LF/HF; increased sleep fragmentation [10,11,31] |
If high-arousal readouts persist after controlling pain/medications, the phenotype is supported; otherwise falsified [4,11,31] |
|
“Yin-predominant / low arousal / low metabolic state” |
N/E (brain network + stress axis) |
Tendency toward EEG burst suppression; reduced temperature/metabolic rate; blunted or delayed stress-hormone responses [11,13] |
If a persistently low-reactivity trajectory remains under matched depth and stable physiology, it can serve as a testable phenotype [11,33] |
|
“Yin-Yang reconstruction / rebalancing” |
N/A/E/I (multi-axis) |
HRV recovery speed + inflammatory decay speed (IL-6/CRP) + EEG complexity rebound trajectory + PROs (sleep/fatigue) [10,11,31,32,36-38] |
If an intervention improves these trajectories in concert and aligns with better outcomes, support; otherwise not supported [31,32,35,36-38] |
|
“Rising-falling / transitions” |
Time-window concept (perioperative windowing) |
Windowed trajectories across preop-induction-maintenance-emergence-PACU-POD1-3 [31,32,34,36-38] |
Only use the term if directional changes occur within prespecified windows and are reproducible [31,32,34,36-38] |
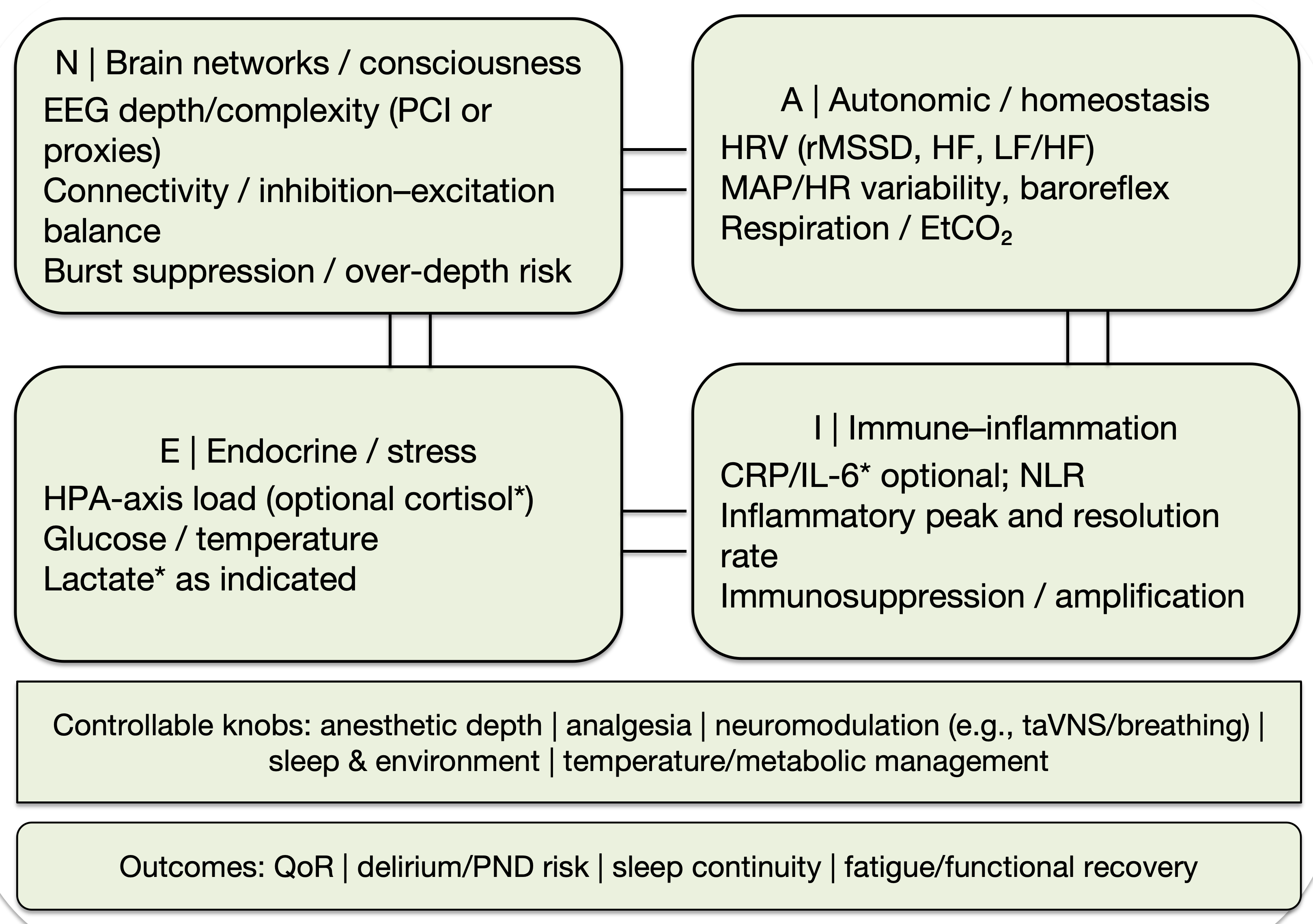
Figure 1: Conceptual NAEI network for multi-scale perioperative tuning: measurable anchors, controllable levers, and outcomes.
Each module is paired with representative measurable anchors and corresponding controllable intervention knobs (e.g., anesthetic depth, analgesia, neuromodulation such as taVNS/breath training, sleep and environmental optimization, temperature/metabolic management). Conceptual schematic; not a mechanistic or quantitative model.
Abbreviations: A, autonomic/homeostasis; CRP: C-reactive protein; EEG: Electroencephalography; EtCO₂: End-Tidal Carbon Dioxide; HPA: Hypothalamic Pituitary Adrenal; HF: High Frequency; HR: Heart Rate; HRV: Heart Rate Variability; I: immune Inflammation; IL-6: Interleukin-6; LF: Low Frequency; MAP: Mean Arterial Pressure; N: Brain Networks/Consciousness; NAEI: Neural Autonomic Endocrine Immune; NLR: Neutrophil to Lymphocyte Ratio; PCI: Perturbational Complexity Index; PND: Perioperative Neurocognitive Disorders; QoR: Quality of Recovery; rMSSD: Root Mean Square of Successive Differences; taVNS: Transcutaneous Auricular Vagus Nerve Stimulation.
Anesthetic techniques and cancer outcomes
In oncologic surgery, perioperative immunosuppression is widely regarded as a critical window relevant to recurrence and metastasis. Anesthetic and surgical techniques may modulate the tumor microenvironment through effects on neuroendocrine stress signaling and immune cell function, including NK activity and T-cell differentiation [33,34]. The autonomic and immune systems also interact bidirectionally via neuro-immune reflex pathways [34,35].
Randomized trials in breast cancer surgery comparing combined regional anesthesia-analgesia plus general anesthesia versus general anesthesia alone found no significant difference in long-term recurrence, although regional techniques can reduce opioid exposure and improve early recovery [36,38-41]. Observational studies report associations between anesthetic technique and long-term outcomes but are constrained by confounding and heterogeneity [37,38]. Recent reviews conclude that current evidence does not support a definitive claim that any anesthetic technique reduces cancer recurrence, while perioperative immune-inflammatory modulation remains a research priority [34,38]. Accordingly, we use conditional, non-causal language and decompose this domain into measurable anchors (inflammation, stress markers, HRV, analgesic quality) and testable pathways (e.g., mediation analyses, embedded randomization, multicenter external validation) [31,32,42,43].
Evolution of Perioperative Medicine and the Role of the Anesthesiologist
Within the perioperative medicine paradigm, the anesthesiologist’s role has expanded from intraoperative drug administration to longitudinal risk management spanning preoperative assessment, intraoperative monitoring, and postoperative recovery [39,40]. Contemporary reviews and ERAS-oriented consensus statements emphasize integrated care across time windows, including multimodal analgesia and opioid minimization, organ protection, early mobilization, nutritional support, and sleep/circadian preservation [40-42]. Perioperative precision medicine further advocates integrating omics, imaging-derived phenotypes, and large-scale data to individualize strategies based on risk phenotypes and network states [40,43]. Within our framework, anesthesiologists can be re-envisioned as “physiological tuners” of the NAEI network: the objective extends beyond achieving anesthetic depth to optimizing multi-axis trajectories across perioperative phases to reduce complications and improve recovery quality [41-43].
Nonpharmacologic Neuromodulation: VNS and Electroacupuncture as Tuning Tools
Nonpharmacologic neuromodulation, including transcutaneous auricular vagus nerve stimulation (taVNS), offers additional tools for NAEI tuning [44,45]. Existing studies suggest VNS-related approaches may influence susceptibility to cardiac arrhythmias (including atrial fibrillation) via autonomic rebalancing, but observed effects depend on stimulation parameters, indications, and study design [35,44]. In healthy participants and in patients with rheumatoid arthritis or systemic lupus erythematosus, deep-breathing protocols combined with taVNS have produced measurable HRV changes, supporting the feasibility of modulating slower dynamics along the vagal-immune/inflammatory axis [31,45].
We therefore position taVNS and electroacupuncture as “parallel tuners” at the systems-network level (Figure 2). In this framework, these interventions primarily target slower dynamics within the autonomic (A) and immune-inflammatory (I) modules and are evaluated jointly with perioperative outcomes. In perioperative settings, evidence also supports measurable links between electroacupuncture, inflammatory markers, and neurocognitive endpoints in older adults [32,46]. Accordingly, pharmacologic anesthesia provides rapid, predictable control of consciousness and analgesia, whereas nonpharmacologic tuners are modeled as adjuncts that may reshape sympatho-vagal balance and immune-inflammatory load over longer time scales, potentially influencing PND, infection risk, and recovery trajectories [32,41,44,45].
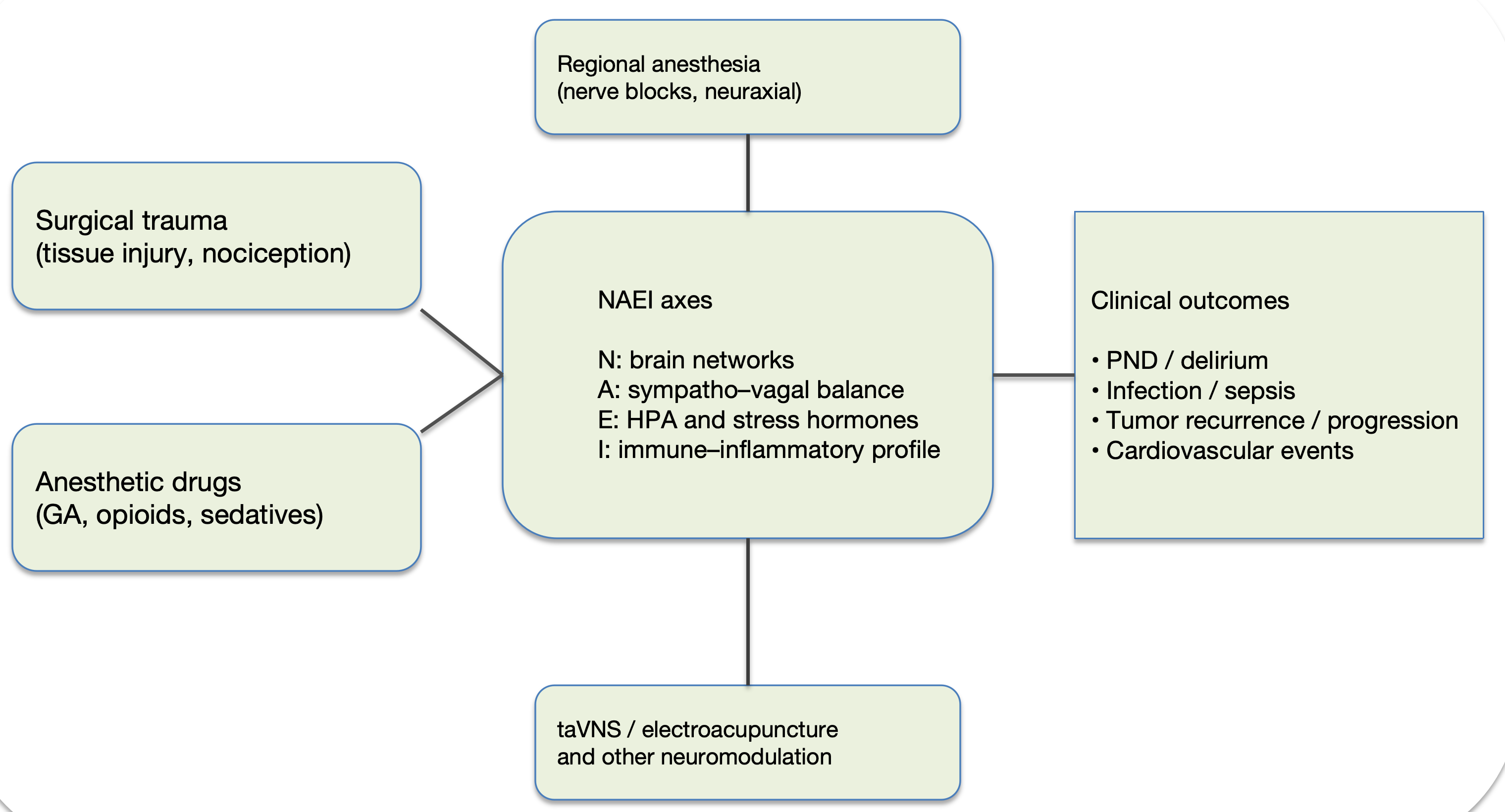
Figure 2: Input locations of non‑pharmacologic neuromodulation (taVNS/electroacupuncture) within the NAEI network and links to outcomes.
Illustrates how surgical trauma, anesthetic drugs and techniques (including regional anesthesia), and non‑pharmacologic neuromodulation (taVNS/electroacupuncture) can act as parallel tuners on the NAEI axes, reshaping perioperative trajectories and downstream outcomes (e.g., PND/delirium, infection/sepsis, cancer recurrence/progression, cardiovascular events). Conceptual schematic intended to clarify the “inputs → axes → outcomes” chain.
Abbreviations: A: Autonomic (sympatho-vagal) Balance; E: Endocrine (hypothalamic-pituitary-adrenal, HPA: Axis and Stress Hormones); GA: General Anesthesia; HPA: Hypothalamic Pituitary Adrenal; I: Immune Inflammatory profile; N: Brain Networks; NAEI: Neural Autonomic Endocrine Immune; PND: Perioperative Neurocognitive Disorders; taVNS: Transcutaneous Auricular Vagus Nerve Stimulation
The Micro Level: Yin Yang and Quantum Concepts as a Hypothesis Generating Layer
This section treats Yin-Yang and quantum-related concepts strictly as cross-disciplinary heuristics for hypothesis generation. They are not presented as established mechanisms of anesthesia or consciousness. Any micro-level proposition must be evaluated through measurable proxies and independent evidence at the clinical-operational and systems-network layers.
Microtubule and quantum-related theories: Conditional discussion and falsifiable predictions
The Orch-OR framework proposes that consciousness may involve quantum processes within neuronal microtubules and that anesthetics could disrupt these processes [47,48]. Molecular modeling and theoretical work further suggests that some inhalational anesthetics may interact with specific tubulin domains, potentially altering microscopic energy or information channels; here, such claims are treated as testable hypotheses requiring independent empirical validation rather than established mechanisms [48-50]. CaMKII-mediated microtubule phosphorylation and cytoskeletal signaling have been implicated in memory-related processes, but substantial multilevel evidence is required to connect these findings to clinical anesthesia phenomena [13,49]. More broadly, quantum-biology reviews cite examples (e.g., photosynthesis, olfaction) to illustrate potential roles for coherence or tunneling; these examples do not imply that consciousness is a quantum phenomenon, but they can motivate falsifiable anesthesia-relevant questions [50]. Throughout, micro-level hypotheses are presented alongside non-quantum alternatives, including receptor-synapse-network coupling, cytoskeletal dynamics, and network criticality [13,22,50].
Yin yang balance and the oxygen pathway hypothesis: From heuristics to measurable variables
From an energy-metabolism perspective, the “oxygen pathway perturbation” hypothesis proposes that anesthesia may alter oxygen delivery or utilization, thereby influencing cellular energy states relevant to state maintenance [13,51]. Drawing on Yin-Yang concepts as an auxiliary clinical heuristic, we use “Yang” to index outward, high-activity/high-complexity/high-metabolic configurations and “Yin” to index inward, stable, low-noise, restorative configurations. These descriptors are mapped to measurable variables (e.g., HRV, hormonal markers, immune indicators) rather than treated as mechanistic explanations [31,52].
To minimize abstraction, we apply a “parallel description” rule: whenever Yin/Yang, ascent/descent, balance/imbalance, or transition/reconstruction terms are used, at least one measurable proxy (EEG, HRV, inflammation, metabolism, or behavioral outcome) is stated alongside the label, ensuring falsifiability and reproducibility [11,31,35].
Operational definition (heuristic, falsifiable): here, “Yin-Yang reconfiguration” denotes a time-windowed, reversible shift of the perioperative trajectory from a high-stress/low-stability configuration toward a more homeostatic, recovery-supportive configuration, expressed only via measurable proxies [31,35]. A “reconfiguration event/trajectory” is recorded when (i) brain-state readouts move toward recovery-consistent patterns across emergence-POD1 (e.g., reduced burst-suppression burden and/or recovery of interpretable EEG complexity/connectivity markers) [11,17-19]; (ii) autonomic balance improves under stable ventilation/hemodynamics (e.g., increased rMSSD/HF in short rolling windows) [31,35]; and (iii) stress-inflammatory proxies trend toward lower load where feasible (e.g., IL-6/CRP and/or cortisol), accompanied by improved clinical anchors (e.g., QoR, sleep continuity) [32,41]. Falsification: if these proxies show no incremental explanatory value beyond anesthetic/analgesic exposure and standard physiologic covariates, or fail to replicate across prespecified windows/cohorts, the mapping is rejected [22].
Correspondences between Yin-Yang descriptors and measurable anchors are aligned in (Figure 3). Abstract terms are mapped to NAEI nodes and a minimal indicator set, with time-window-specific priority actions and reassessment points detailed in (Table 5).
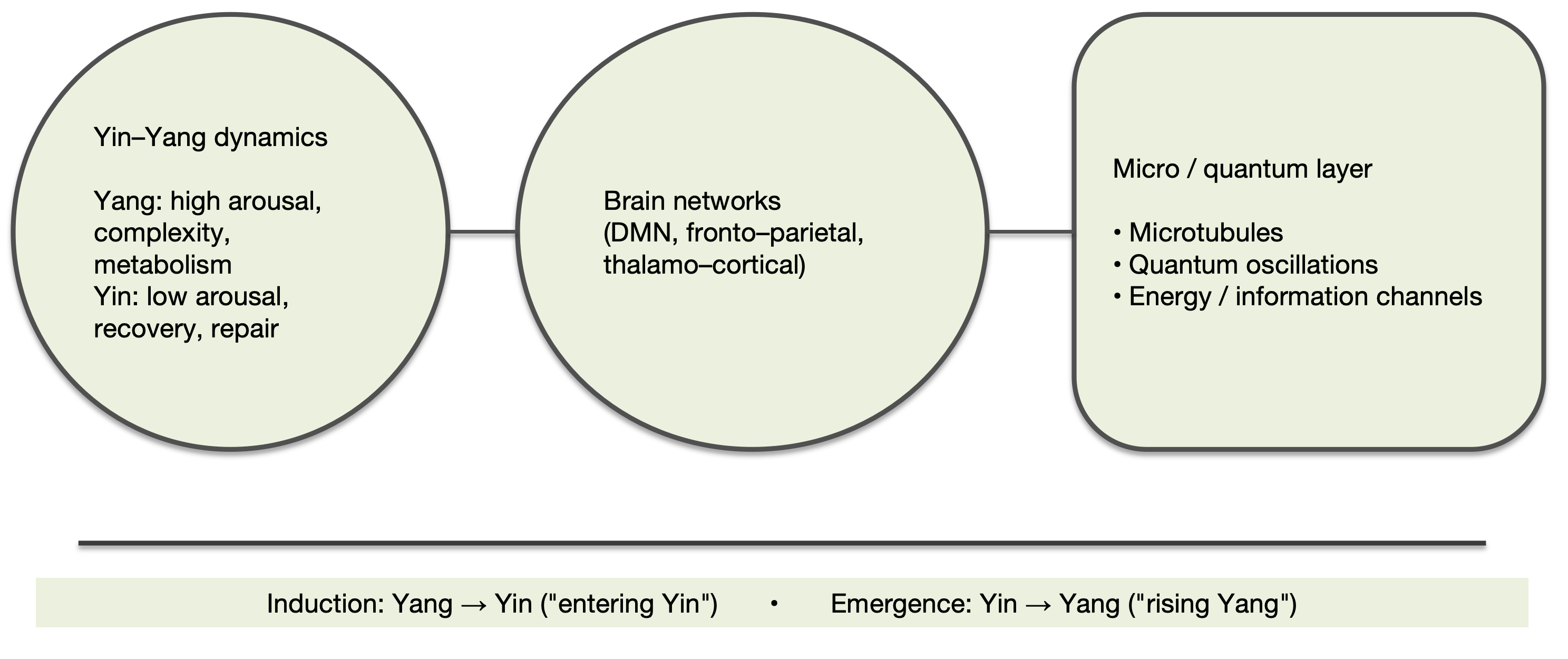
Figure 3: Cross‑scale analogy for the hypothesis‑generation layer: Yin-Yang dynamics, brain networks, and candidate micro‑level/quantum hypotheses.
Uses Yin-Yang dynamics strictly as a heuristic, cross‑disciplinary language to relate brain‑network states (e.g., DMN, frontoparietal networks, thalamo‑cortical pathways) to candidate micro‑level/quantum hypotheses (e.g., microtubules, quantum oscillations, and energy/information channels).
The label ‘hypothesis‑generating’ denotes hypothesis generation only and does not imply mechanistic identity. Any clinically relevant claim must be tested through measurable higher‑level proxies and independent empirical studies.
Abbreviations: DMN: Default Mode Network; Yin-Yang, traditional Chinese medicine framework describing complementary low- and high-arousal states (Yin: low arousal, recovery, repair; Yang: high arousal, complexity, metabolism).
Table 5: Operationalizing Yin-Yang terms: perioperative time windows with a minimal measurement set and reassessment windows (heuristic → falsifiable proxies; for clinical and study design use).
|
Time window |
Primary risks/goals (examples) |
Minimal measurement set (examples) |
Priority actions and reassessment window (examples) |
|
Preoperative baseline |
Risk stratification; baseline anxiety/sleep/inflammation |
Baseline HRV; sleep scales/history; CRP/IL-6 (optional) [31,32] |
Set individualized target ranges; preop sleep/anxiety intervention (as feasible) [40,43] |
|
Induction |
Rapidly reach target state; avoid excessive depth |
EEG (raw waveform + optional processed index such as BIS/entropy); hemodynamics |
Titrate based on EEG/circulatory responses; reassess every 1–3 min [31,32] |
|
Maintenance |
Maintain stability + adequate analgesia + avoid burst suppression |
EEG waveform (optional complexity); analgesia indicators; HRV trend [10,11,31] |
Optimize analgesia before deepening sedation; reassess every 10–15 min [41,31] |
|
Emergence |
Smooth emergence; reduce agitation/delirium risk |
EEG transition patterns; HRV; pain scores/analgesic needs [10,31] |
Reduce high stress: analgesia/environment/neuromodulation; reassess in PACU [41,44] |
|
PACU (0-2 h) |
Pain-delirium-nausea-temperature management |
Outcome-triggered rescue actions; reassess every 30-60 min [41,31] |
|
|
Postoperative night |
Sleep continuity and control of inflammatory peak |
Sleep-friendly environment + analgesia optimization; reassess next day [40,41] |
|
|
POD1 |
Early mobilization and recovery quality |
Adjust analgesia/rehab according to trajectories; reassess on POD2 [41,43] |
|
|
POD2–3 |
Early detection of complications (infection/PND) |
Temperature/inflammation (as needed); delirium screening; PROs [32,46] |
Trigger further assessment/intervention based on abnormal anchors [43,46] |
Note: Terms such as “Yin/Yang,” “rising/falling,” “balance/imbalance” serve only as indexing labels for clinical narration Their validity must be tested against consistency between proxy trajectories and outcomes; they do not constitute causal claims.
Cellular/Molecular Anesthesiology, Personalization, and Neurotoxicity: Limits of the Evidence and a Research Agenda
Cellular/molecular mechanisms, personalization, and evidence limits
At the cellular and molecular level, “cellular and molecular anesthesiology” emphasizes interpreting anesthetic effects through ion channels, receptors, and signaling pathways, and translating basic discoveries into clinical practice [13,53]. Precision medicine adds a practical requirement: incorporate genomics, interindividual variability in drug response, and ongoing professional education to address heterogeneous risk and variable benefit [54-56].
For anesthetic-related neurotoxicity, bioinformatics and animal models suggest candidate pathways for propofol, including mitochondrial dysfunction, oxidative stress, and apoptosis, but inference is constrained by model heterogeneity and limited clinical comparability [13,55]. Database and large-scale analytic commentaries further note that clinical evidence is often limited by small samples, short follow-up, and residual confounding, supporting the need for structured data platforms that enable reproducible real-world research and risk stratification [43,56].
Pediatric neurodevelopment and long-term evidence gaps
Pediatric and developmental neurobiology remains a major evidence gap. Most data derive from animal work, mechanistic studies, and observational cohorts, and extrapolation to specific drugs, doses, exposure windows, and long-term outcomes is complicated by developmental stage and perioperative confounding; “any anesthesia” exposure definitions are rarely sufficient for causal attribution [55,61]. We therefore do not estimate a definitive effect size. Instead, we propose a pragmatic, time-windowed agenda: prospective cohorts or embedded randomization without added ethical burden; a minimal, age-appropriate anchor set across prespecified perioperative phases-EEG signatures of depth/vulnerability (including burst-suppression burden where relevant), HRV/autonomic stress reactivity (e.g., rMSSD/HF), feasible inflammatory/endocrine readouts (IL-6/CRP, cortisol), and sleep continuity-paired with standardized neurodevelopmental outcomes at harmonized follow-up intervals [31,32,57,62,63]. Analyses should explicitly model surgical stress, analgesia quality, and sleep interventions as key covariates, and use designs that strengthen causal inference (e.g., negative controls/sibling comparisons, preregistered sensitivity analyses) before attributing long-term outcomes to anesthetic drugs alone [56,62,63].
Monitoring and Closed-Loop Control: From EEG to Connectivity and NAEI Trajectories
Early EEG reviews summarized principles and clinical applications of brain monitoring in the operating room and ICU and repeatedly caution that reliance on a single processed index without inspecting the raw waveform can misclassify brain state [57,62]. Subsequent work extends this point in two directions. First, anesthesia-related emergence and postoperative affective experience may involve midbrain-limbic (reward-related) circuitry, motivating broader state models beyond “depth” alone [58,60]. Second, connectivity-focused commentaries argue that functional connectivity and network topology can capture drug-induced network reconfiguration and improve alignment between neurophysiologic readouts and behavioral trajectories [59,63]. Conceptual models such as “cognitive unbinding” similarly propose that anesthesia disrupts effective connectivity across cognitive modules, offering a network-level account of unconscious states [59,60]. Developmental connectivity findings further suggest age-dependent vulnerability and maturation effects, supporting greater caution and longer follow-up in pediatric anesthesia [56,61].
From a technology and evidence perspective, depth-of-anesthesia monitoring reviews highlight persistent limitations in algorithm transparency, generalizability, and outcome-level validation; interpretation should therefore integrate raw EEG and clinical context rather than depend on a single number [57,62]. Consistent with this literature, we outline a device-agnostic conceptual framework for EEG × HRV integration in closed-loop systems [62,63].
Time window structure
Collect data by perioperative phases (preoperative baseline, induction, maintenance, emergence, PACU, postoperative night, POD1-3) [57,63].
Minimal feature set
EEG (δ/α/β power, burst-suppression ratio; optional complexity/entropy); HRV (rMSSD, HF, LF, LF/HF using 1-5 min rolling windows); inflammation/stress (IL-6/CRP, cortisol where feasible); and clinical anchors (pain, delirium screening, QoR, sleep continuity, fatigue/activity) [57,62].
Decision logic
Treat hemodynamics, ventilation, and temperature as hard constraints; when constraints are met, run a soft-constraint loop of “state estimation → knob adjustment → reassessment.” Examples include down-titrating sedative load when EEG suggests excessive depth or burst suppression, and prioritizing analgesia optimization and nonpharmacologic tuners when HRV/inflammation indicates high stress load [62,63].
Explainable output
Prefer auditable rules or shallow interpretable models (threshold rules, decision trees, interpretable linear/GLM models) that generate traceable recommendations rather than black-box dosing directives [62,63].
To make the “measurable readouts-controllable knobs” mapping explicit, we provide an interface table organized by N/A/E/I modules, linking key state variables to measurable anchors and actionable knobs (Table 6). Figure 4 integrates time-window triggers, priority knobs, and reassessment windows as a process overview aligned to (Table 6).
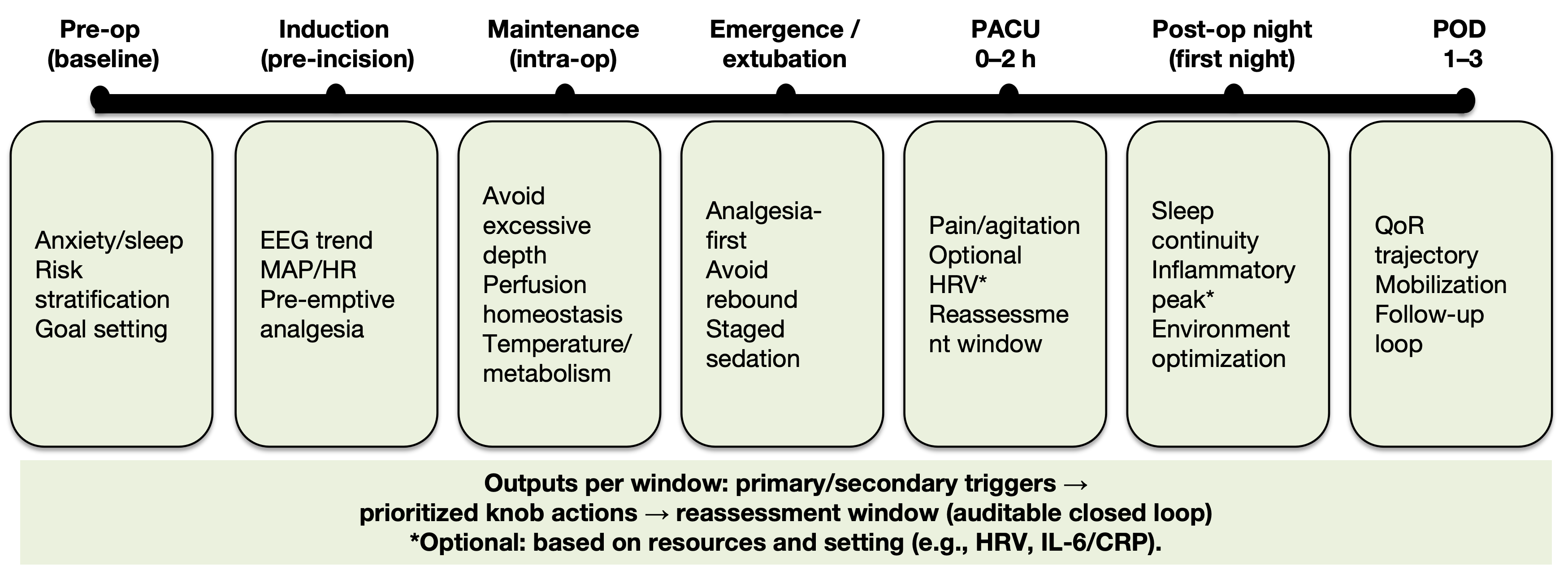
Figure 4: Concept‑level closed‑loop checklist across perioperative time windows: time window-trigger signals-knobs-re‑evaluation interval.
Across key windows (preop baseline, induction, maintenance, emergence, PACU 0-2 h, postoperative night, POD1, POD2-3), the figure summarizes goals/risk points, trigger signals, priority knobs, and re‑evaluation intervals. Together, “measure → adjust → re‑evaluate” forms an auditable tuning loop, translating the readouts-knobs-outcomes relationship into an executable time‑window checklist. Conceptual schematic.
Abbreviations: Pre-op: Preoperative; EEG: Electroencephalography; MAP: Mean Arterial Pressure; HR: Heart Rate; PACU: Post Anesthesia Care Unit; HRV: Heart Rate Variability; POD: Postoperative Day; QoR: Quality of Recovery; IL-6: Interleukin-6; CRP: C-Reactive Protein.
Table 6: Modular interface for closed-loop control: key state variables, measurable anchors, and controllable knobs by N/A/E/I (conceptual).
|
Module |
Key state variables (examples) |
Measurable anchors (examples) |
Controllable knobs (examples) |
|
N (brain network) |
Connectivity, integration, complexity, ignition/broadcast capacity [18,19,22,25] |
EEG spectra/complexity [10,18,19]; TMS-EEG/PCI (optional) [16,63]; fMRI/EEG connectivity (research) [20,21] |
Depth/drug combinations [2,60]; analgesia-sedation balance [41,63]; temperature/CO₂ [13,35]; sleep interventions (context-dependent) [1,22] |
|
A (autonomic) |
Sympatho-vagal balance, reflex sensitivity, cardio-respiratory coupling [31,35] |
HRV [31,45]; hemodynamic reactivity [31,62]; respiratory variability (as feasible) [45,62] |
Analgesia quality/opioid minimization [41,37]; fluids/vasoactives [39,62]; taVNS/TEAS parameters [44,45,46]; respiratory management [45,62] |
|
E (endocrine) |
Cortisol/catecholamines (optional) [33,35]; glycemic variability [33,43] |
Pain and anxiety management [41,43]; anesthetic technique/drug selection [2,37]; circadian/light and sleep [1,40] |
|
|
I (immune-inflammation) |
Inflammatory peak and decay speed; window of immunosuppression [32,34,35] |
Minimally invasive/ERAS [41,40]; opioid minimization [41,37]; neuromodulation [44,45,46]; optimization of analgesia and sleep [41,31] |
Note: “Measurable anchors/controllable knobs” are conceptual examples used to define interfaces and an audit trail Implementation should be tailored to procedure context, risk stratification, and local workflows.
The Three-Layer Multiscale Tuning Model and Working Definition
The structured relationship among the three layers-and the operational “knob-readout-outcome” loop-is summarized in (Figure 5). Based on the evidence reviewed, we conceptualize anesthesia as a multiscale tuning process spanning clinical operations, systems-network physiology, and hypothesis generation:
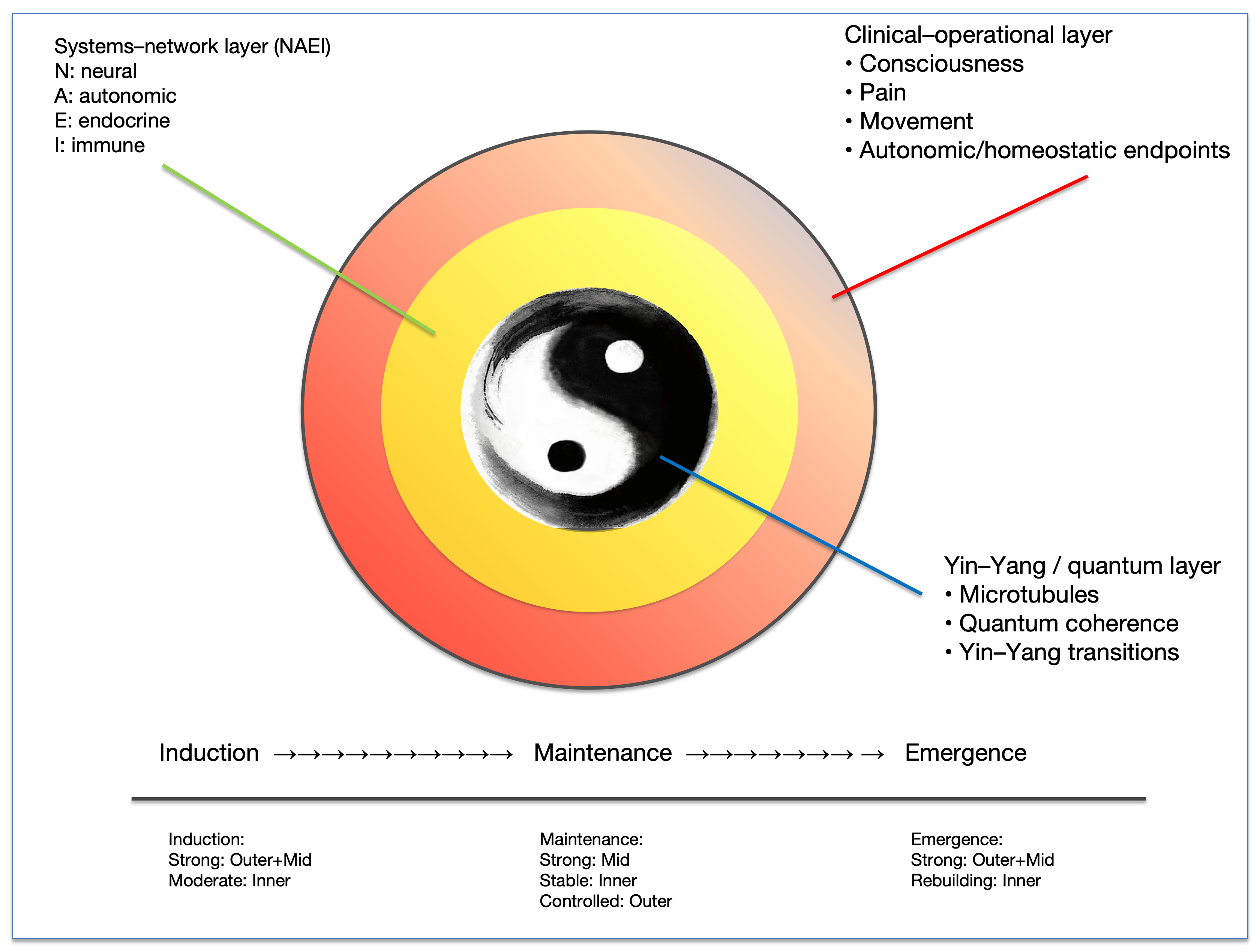
Figure 5: Overall framework of the three‑layer multi‑scale tuning model: clinical-operational layer × system-network layer (NAEI) × hypothesis‑generation layer (Yin-Yang/quantum).
A concentric schematic integrates the three working definitions: the clinical-operational layer (controllable modulation of consciousness, pain, motor output, and homeostatic endpoints), the system-network layer (measurable NAEI readouts and controllable knobs), and the hypothesis‑generation layer (strictly bounded, conditional discussion). The timeline (induction → maintenance → emergence) highlights how the tuning emphasis and controllability shift by phase. Conceptual integration figure.
Abbreviations: NAEI: Neural Autonomic Endocrine Immune Network; N: Neural; A: Autonomic; E: Endocrine; I: Immune; Yin-Yang, conceptual balance between low-arousal/restorative (Yin) and high-arousal/active (Yang) states; Induction, anesthetic induction phase; Maintenance, intraoperative maintenance phase; Emergence, recovery from anesthesia phase.
Clinical-operational layer
Using drugs and/or neuromodulation, anesthesia enables multi-target state control of consciousness, pain, motor output, and autonomic responses, allowing invasive procedures to proceed safely with manageable stress load [1-3,9,10,39,41].
Systems-network layer (NAEI)
Anesthesia and perioperative interventions shape large-scale brain connectivity, sympatho-vagal balance, hormonal dynamics, and immune-inflammatory profiles across time windows; these trajectories show testable associations with PND, infection, recovery patterns, and selected longer-term outcomes [6-8,16-21,31-38,43-46].
Hypothesis-generating layer (Yin-Yang/quantum)
Cross-disciplinary micro-level concepts are used only to generate falsifiable questions and do not replace mainstream receptor-synapse-circuit-network explanations or support clinical recommendations [47-52].
Working definition
Anesthesia is a multiscale tuning process induced by drugs and/or neuromodulation and reversibly terminated. Clinically, it is multi-target state control of consciousness, analgesia, motor output, and autonomic responses. Systemically, it modulates the NAEI network across perioperative time windows to maintain or optimize homeostasis and recovery-relevant outcomes. At the micro-level, controversial cross-disciplinary concepts serve only as heuristic language for testable questions anchored to measurable proxies [1-3,31,32,47-52].
Limitations and Future Directions
This work is a narrative synthesis and framework paper; we did not follow a PRISMA workflow or perform quantitative pooling, so selection and publication bias remain possible. We mitigated these risks through evidence-gradient labeling, explicit measurable anchors, and cross-disciplinary citation tracing [41,43]. Clinical evidence in heterogeneous domains (e.g., anesthesia-cancer links, neurotoxicity) is constrained by complex confounding; we therefore provide an organizing framework rather than causal claims [34,36-38]. Controversial micro-level theories lack strong empirical support and are restricted to hypothesis generation, presented alongside alternative explanations and falsifiable testing paths [1-3,31,22,47-52].
Future work should prioritize outcome-anchored questions (PND, infection, recovery trajectories, cancer-related outcomes) and collect a minimal indicator set (EEG/HRV/inflammation/sleep/PRO) within feasible time windows, with multicenter external validation. This platform can then support focused systematic reviews/meta-analyses and prospective trials built around specific, auditable anchor questions [31,32,41,43,63].
Conclusion
As perioperative precision medicine converges with modern consciousness science, we propose reframing anesthesia from “drug-induced reversible unconsciousness” into an auditable multiscale tuning process. The clinical-operational layer specifies controllable knobs that deliver safety and comfort, while the systems-network layer uses NAEI trajectories to explain variability in recovery. A minimal set of measurable anchors links state, intervention, and outcome into a re-evaluable loop across defined perioperative windows and across centers [1-3,31,32,41,43,63].
This framework does not claim new causal conclusions. Instead, it provides a working language that can be supported-or rejected-by data: it constrains interpretation with evidence gradients, expresses hypotheses in falsifiable proxy terms, and favors external validation over single-center extrapolation [31,32,41,43]. The highest-yield next step is to focus on key endpoints (PND, infection, recovery trajectories, cancer-related outcomes), collect a minimal indicator set (EEG/HRV/inflammation/sleep/PRO) within prespecified windows, and use embedded trials plus multicenter validation to translate multiscale tuning from concept to a testable perioperative research and practice paradigm [31,32,36-38,41,43,63].
Funding
National Natural Science Foundation of China (Project Approval Number: 81200858); Jiangsu Province 333 High-level Talent Training Project [Certificate No.: (2022) No. 3-10-007].
Clinical Trials from Nanjing Drum Tower Hospital, Affiliated Hospital of Medical School, Nanjing University, the Huai'an Matching Assistance Special Project (2024-2025).
Conflict of Interest
None.
AI assisted Declaration
The authors used generative AI tools (e.g., ChatGPT, OpenAI) to assist in language polishing and formatting. All intellectual content, analyses, and conclusions are solely those of the authors.”
Presentation
None declared.
References
- Brown EN, Lydic R, Schiff ND. General anesthesia, sleep, and coma. N Engl J Med. 2010;363(27):2638-2650. doi:10.1056/NEJMra0808281.
- Brown EN, Purdon PL, Van Dort CJ. General anesthesia and altered states of arousal: a systems neuroscience analysis. Annu Rev Neurosci. 2011;34:601-628. doi:10.1146/annurev-neuro-060909-153200.
- Alkire MT, Hudetz AG, Tononi G. Consciousness and anesthesia. Science. 2008;322(5903):876-880. doi:10.1126/science.1149213.
- Montupil J, Cardone P, Staquet C, Bonhomme A, Defresne A, Martial C, et al. The nature of consciousness in anaesthesia. BJA Open. 2023;8:100224. doi:10.1016/j.bjao.2023.100224. PMID:37780201.
- Bonhomme V, Staquet C, Montupil J, Defresne A, Kirsch M, Martial C, et al. General anesthesia: a probe to explore consciousness. Front Syst Neurosci. 2019;13:36. doi:10.3389/fnsys.2019.00036.
- Hudetz AG. General anesthesia and human brain connectivity. Brain Connect. 2012;2(6):291-302. doi:10.1089/brain.2012.0107.
- Hudetz AG, Mashour GA. Disconnecting consciousness: is there a common anesthetic endpoint? Anesth Analg. 2016;123(5):1228-1240. doi:10.1213/ANE.0000000000001353
- Mashour GA, Hudetz AG. Neural correlates of unconsciousness in large-scale brain networks. Trends Neurosci. 2018;41(3):150-160. PMID:29409683.
- Lewis LD, Weiner VS, Mukamel EA, Donoghue JA, Eskandar EN, Madsen JR, et al. Rapid fragmentation of neuronal networks at the onset of propofol-induced unconsciousness. Proc Natl Acad Sci U S A. 2012;109(49):E3377-E3386.
- Purdon PL, Pierce ET, Mukamel EA, Prerau MJ, Walsh JL, Wong KFK, et al. Electroencephalogram signatures of loss and recovery of consciousness from propofol. Proc Natl Acad Sci U S A. 2013;110(12):E1142-E1151.
- Moody OA, Zhang ER, Vincent KF, Kato R, Melonakos ED, Nehs CJ, et al. The neural circuits underlying general anesthesia and sleep. Anesth Analg. 2021;132(5):1254-1264.
- Yi R, Cheng S, Zhong F, Luo D, You Y, Yu T, et al. GABAergic neurons of anterior thalamic reticular nucleus regulate states of consciousness in propofol- and isoflurane-mediated general anesthesia. CNS Neurosci Ther. 2024;30:e14782. doi:10.1111/cns.14782. PMID:38828651.
- Hao X, Ou M, Zhang D, Zhao W, Yang Y, Liu J, et al. The effects of general anesthetics on synaptic transmission. Curr Neuropharmacol. 2020;18(10):936-965. PMID:32106800.
- Forman SA, Chin VA. General anesthetics and molecular mechanisms of unconsciousness. Int Anesthesiol Clin. 2008;46(3):43-53. doi:10.1097/AIA.0b013e3181755da5. PMID:18617817.
- Sanders RD, Tononi G, Laureys S, Sleigh JW. Unresponsiveness ≠ unconsciousness. Anesthesiology. 2012;116(4):946-959. doi:10.1097/ALN.0b013e318249d0a7. PMID:22314293.
- Casali AG, Gosseries O, Rosanova M, Boly M, Sarasso S, Casali KR, et al. A theoretically based index of consciousness independent of sensory processing and behavior. Sci Transl Med. 2013;5(198):198ra105.
- Lee M, Sanz LRD, Barra A, Bourdillon P, Sitt JD, Naccache L, et al. Quantifying arousal and awareness in altered states of consciousness using interpretable deep learning. Nat Commun. 2022;13:1064.
- Kim H, Hudetz AG, Lee J, Mashour GA, Lee U. Estimating the integrated information measure phi from high-density electroencephalography during states of consciousness in humans. Front Hum Neurosci. 2018;12:42.
- Blain-Moraes S, Tarnal V, Vanini G, Alexander A, Rosen D, Shortal B, et al. Network efficiency and posterior alpha patterns are markers of recovery from general anesthesia: a high-density electroencephalography study in healthy volunteers. Front Hum Neurosci. 2017;11:328.
- Bonhomme V, Vanhaudenhuyse A, Demertzi A, Bruno MA, Jaquet O, Bahri MA, et al. Resting-state network-specific breakdown of functional connectivity during ketamine alteration of consciousness in volunteers. Anesthesiology. 2016;125(5):873-888. PMID:27496657.
- Luppi AI, Golkowski D, Ranft A, Ilg R, Jordan D, Bzdok D, et al. General anaesthesia decreases the uniqueness of brain functional connectivity across individuals and species. Nat Hum Behav. 2025;9(5):987-1004. doi:10.1038/s41562-025-02121-9.
- Seth AK, Bayne T. Theories of consciousness. Nat Rev Neurosci. 2022;23(7):439-452. doi:10.1038/s41583-022-00587-4. PMID:35505255.
- Boly M, Massimini M, Tsuchiya N, Postle BR, Koch C, Tononi G. Are the neural correlates of consciousness in the front or in the back of the cerebral cortex? J Neurosci. 2017;37(40):9603-9613. doi:10.1523/JNEUROSCI.3218-16.2017. PMID:28978697.
- Northoff G, Lamme V. Neural signs and mechanisms of consciousness: is there a potential convergence of theories of consciousness in sight? Neurosci Biobehav Rev. 2020;118:568-587. doi:10.1016/j.neubiorev.2020.07.019. PMID:32783969.
- Mashour GA, Roelfsema P, Changeux JP, Dehaene S. Conscious processing and the global neuronal workspace hypothesis. Neurosci Conscious. 2020;2020(1):niaa010. doi:10.1093/nc/niaa010.
- Demertzi A, Tagliazucchi E, Dehaene S, Deco G, Barttfeld P, Raimondo F, et al. Human consciousness is supported by dynamic complex patterns of brain signal coordination. Sci Adv. 2019;5(2):eaat7603. doi:10.1126/sciadv.aat7603.
- Vanhaudenhuyse A, Demertzi A, Schabus M, Noirhomme Q, Bredart S, Boly M, et al. Two distinct neuronal networks mediate the awareness of environment and of self. J Cogn Neurosci. 2011;23(3):570-578.
- Crick F, Koch C. Towards a neurobiological theory of consciousness. Semin Neurosci. 1990;2(5):263-275.
- Fazekas P, Overgaard M. Perceptual consciousness and cognitive access: an introduction. Philos Trans R Soc Lond B Biol Sci. 2018;373(1755):20170340. doi:10.1098/rstb.2017.0340.
- Noel JP, Ishizawa Y, Patel SR, Eskandar EN, Wallace MT. Leveraging nonhuman primate multisensory neurons and circuits in assessing consciousness theory. J Neurosci. 2019;39(38):7485-7500.
- Wujtewicz M, Owczuk R. Heart rate variability in anaesthesiology – narrative review. Anaesthesiol Intensive Ther. 2023;55(1):1-8. doi:10.5114/ait.2023.126309. PMID:37306266.
- O’Bryan LJ, Atkins KJ, Lipszyc A, Scott DA, Silbert BS, Evered LA. Inflammatory biomarker levels after propofol or sevoflurane anaesthesia: a systematic review and meta-analysis. Anesth Analg. 2022;134(1):69-81. doi:10.1213/ANE.0000000000005671. PMID:34908547.
- Kim R. Effects of surgery and anesthetic choice on immunosuppression and cancer recurrence. J Transl Med. 2018;16(1):8.
- Horowitz M, Neeman E, Sharon E, Ben-Eliyahu S. Exploiting the critical perioperative period to improve long-term cancer outcomes. Nat Rev Clin Oncol. 2015;12(4):213-226.
- Kenney MJ, Ganta CK. Autonomic nervous system and immune system interactions. Compr Physiol. 2014;4(3):1177-1200.
- Sessler DI, Pei L, Huang Y, Fleischmann E, Marhofer P, Kurz A, et al. Recurrence of breast cancer after regional or general anesthesia: a randomized controlled trial. Lancet. 2019;394(10211):1807-1815. doi:10.1016/S0140-6736(19)32313-X.
- Makito K, Matsui H, Fushimi K, Yasunaga H. Volatile versus total intravenous anesthesia for cancer surgery: a propensity-matched analysis. Anesthesiology. 2020;133(4):764-773. doi:10.1097/ALN.0000000000003440.
- Ahn HJ. Anesthesia and cancer recurrence: a narrative review. Anesth Pain Med (Seoul). 2024;19(2):94-108.
- Wacker J, Staender S. The role of the anesthesiologist in perioperative patient safety. Curr Opin Anaesthesiol. 2014;27(6):649-656.
- Moningi S, Patki A, Padhy N, Ramachandran G, Choudhury M. Enhanced recovery after surgery: An anesthesiologist’s perspective.J Anaesthesiol Clin Pharmacol. 2019;35(Suppl 1):S5–S13.
- McEvoy MD, Scott MJ, Gordon DB, Grant SA, Thacker JK, Wu CL, et al. American Society for Enhanced Recovery and Perioperative Quality Initiative joint consensus statement on optimal analgesia within an enhanced recovery pathway for colorectal surgery. Perioper Med (Lond). 2017;6:8.
- Rock P. The future of anesthesiology is perioperative medicine. Anesthesiol Clin North Am. 2000;18(3):495-513. doi:10.1016/S0889-8537(05)70176-0. PMID:10989705.
- Pal N, Kertai MD. Perioperative precision medicine: where are we in 2020? Curr Opin Anaesthesiol. 2020;33(3):463-471.
- Zafeiropoulos S, et al. Autonomic neuromodulation for atrial fibrillation after cardiac surgery: JACC review topic of the week. J Am Coll Cardiol. 2022;79(7):682-694. doi:10.1016/j.jacc.2021.12.010. PMID:35177198.
- Jensen MSK, Liboriussen CH, Andersen SS, Kristensen SL, et al. Modulating heart rate variability through deep breathing exercises and transcutaneous auricular vagus nerve stimulation: a study in healthy participants and in patients with rheumatoid arthritis or systemic lupus erythematosus. Sensors (Basel). 2022;22(20):7884. doi:10.3390/s22207884.
- Ye L, Hong L, Li J, Xue R, Liu Y, Wang Y, et al. Electroacupuncture for postoperative cognitive dysfunction: a systematic review and meta-analysis protocol. Medicine (Baltimore). 2021;100(4):e23891. doi:10.1097/MD.0000000000023891.
- Hameroff S, Penrose R. Consciousness in the universe: a review of the ‘Orch OR’ theory. Phys Life Rev. 2014;11(1):39-78.
- Craddock TJ, Hameroff SR, Ayoub AT, Klobukowski M, Tuszynski JA. Anesthetics act in quantum channels in brain microtubules to prevent consciousness. Curr Top Med Chem. 2015;15(6):523-533.
- Craddock TJ, Tuszynski JA, Hameroff S. Cytoskeletal signaling: Is memory encoded in microtubule lattices by CaMKII phosphorylation?PLoS Comput Biol. 2012;8(3):e1002421. doi:10.1371/journal.pcbi.1002421.
- Lambert N, Chen YN, Cheng YC, Li CM, Chen GY, Nori F. Quantum biology. Nat Phys. 2013;9(1):10-18.
- Hu H, Wu M. Mechanism of anesthetic action: oxygen pathway perturbation hypothesis. Med Hypotheses. 2001;57(3):357-363.
- Yin Q, Wang JF, Cheng HC, Cheng MY, Cheng W. Yin-Yang balance concepts for clinical diagnosis and treatment: an integrated view of Chinese medicine, anesthesiology, and quantum mechanics. Transl Perioper Pain Med. 2024;11(1):560-573.
- Dabbagh A, Elyassi H. Cellular and molecular anesthesia: from bench to bedside.J Cell Mol Anesth.2016;1(1):e147826.
- Sezari P, Dabbagh A. Personalized medicine: the paradigm shift in medicine mandating lifelong learning. J Cell Mol Anesth. 2019;4:31-32.
- He T, Huang J, Peng B, Wang M, Shui Q, Cai L. Screening of potential biomarkers in propofol-induced neurotoxicity via bioinformatics prediction and experimental verification. Am J Transl Res. 2024;16(3):755-767.
- Obara S. Challenges in database research for anesthetic neurotoxicity. J Anesth. 2025;39(2):321-322.
- Freye E, Levy JV. Cerebral monitoring in the operating room and the intensive care unit: an introductory for the clinician and a guide for the novice wanting to open a window to the brain. Part I: the electroencephalogram. J Clin Monit Comput. 2005;19(1-2):1-76. PMID:16167222.
- Heshmati M, Bruchas MR. Historical and modern evidence for the role of reward circuitry in emergence. Anesthesiology. 2022;136(6):997-1014.
- Ramani R. Connectivity. Curr Opin Anaesthesiol. 2015;28(5):498-504. doi:10.1097/ACO.0000000000000237. PMID:26270569.
- Mashour GA. Cognitive unbinding: a neuroscientific paradigm of general anesthesia and related states of unconsciousness. Neurosci Biobehav Rev. 2013;37(10 Pt 2):2751-2759.
- Chen X, Zheng X, Cai J, Yang X, Lin Y, Wu M, et al. Effect of anesthetics on functional connectivity of developing brain. Front Hum Neurosci. 2022;16:853816.
- Bruhn J, Myles PS, Sneyd R, Struys MM. Depth of anaesthesia monitoring: what’s available, what’s validated and what’s not? Br J Anaesth. 2006;97(1):85-94.
- Purdon PL, Sampson A, Pavone KJ, Brown EN. Clinical electroencephalography for anesthesiologists: Part I: Background and basic signatures.Anesthesiology. 2015;123(4):937-960. doi:10.1097/ALN.0000000000000841. PMID:26275092.
Table of Contents
- Abstract
- Keywords
- Introduction
- Methods Overview: A Minimal, Auditable Strategy for a Narrative Review
- The Sleep Anesthesia Coma Continuum and Phenotypes of Unconsciousness
- Integrating Consciousness Theories with Anesthetic States: Testable Predictions and Contrasts
- The Systems Network Layer: The NAEI Network from Axes to Outcomes
- Evolution of Perioperative Medicine and the Role of the Anesthesiologist
- Nonpharmacologic Neuromodulation: VNS and Electroacupuncture as Tuning Tools
- The Micro Level: Yin Yang and Quantum Concepts as a Hypothesis Generating Layer
- Cellular/Molecular Anesthesiology, Personalization, and Neurotoxicity: Limits of the Evidence and a Research Agenda
- Monitoring and Closed-Loop Control: From EEG to Connectivity and NAEI Trajectories
- The Three-Layer Multiscale Tuning Model and Working Definition
- Limitations and Future Directions
- Conclusion
- Funding
- Conflicts of Interest
- Funding
- AI Use Statement
- Figure 1
- Figure 2
- Figure 3
- Figure 4
- Table
- Table
- References