Translational Perioperative and Pain Medicine (ISSN: 2330-4871)
ARTICLE DOI: 10.31480/2330-4871/213
Research Article | Volume 12 | Issue 1 Open Access
Phenotype-Guided Diagnosis and Intervention Strategies for Perioperative Sleep Disturbances: A Pathway Framework Integrating the CST-DS Axis (Part II)
Qin Yin1#, Fei-Fei Lu2#, Ming-Yue Cheng1, Yu-E Sun3, Jin-Feng Wang4* and Wei Cheng5*
1The Affiliated Hospital of Xuzhou Medical University, Xuzhou 221002, P.R. China
2The TCM Hospital Affiliated to Nanjing University of Chinese Medicine, Xuzhou 221009
3The Affiliated Hospital of Nantong University, No. 20 Xisi Road, Nantong, China
4The Suzhou Hospital of the Chinese Academy of Traditional Chinese Medicine - Xiyuan Hospital, Suzhou, China
5The Affiliated Huai'an No.1 People's Hospital of Nanjing Medical University, Huai'an 223300, P.R. China
#Contributed equally to this work
Wei Cheng, Huai'an No.1 People's Hospital of Nanjing Medical University, Huai'an 223300, China, Tel: +8618796205791, E-mail: 53974314@qq.com; Jin-Feng Wang, The Suzhou Hospital of the Chinese Academy of Traditional Chinese Medicine - Xiyuan Hospital, P.R. Suzhou, China, Tel: +8618168779112, E-mail: 709034013@qq.comEditor: Renyu Liu, MD; PhD; Professor, Department of Anesthesiology and Critical Care, Perelman School of Medicine at the University of Pennsylvania, Center of Penn Global Health Scholar, 336 John Morgan building, 3620 Hamilton Walk, Philadelphia, PA 19104, USA, Fax: 2153495078, E-mail: RenYu.Liu@pennmedicine.upenn.edu
Received: Nov 17, 2025 | Accepted: Jan 14, 2026 | Published: Jan 17, 2026
Citation: Yin Q, Lu FF, Cheng MY, et al. Phenotype-Guided Diagnosis and Intervention Strategies for Perioperative Sleep Disturbances: A Pathway Framework Integrating the CST-DS Axis (Part II). Transl Perioper Pain Med 2026; 12(1):819-830
Abstract
Objective: Building on Part I, Part II operationalizes the framework into a pathway prototype structured as “time window × phenotype × modules” for perioperative assessment and intervention, targeting anesthesiology, sleep medicine, and rehabilitation teams. This article presents a conceptual prototype for hypothesis generation and prospective testing/local adaptation; it is not a validated clinical algorithm or fixed decision tree.
Methods: We conducted an evidence-focused narrative synthesis and framework mapping. High-quality systematic reviews/meta-analyses and representative perioperative studies were used as entry evidence, supplemented by targeted searches of PubMed/MEDLINE, Embase, and the Cochrane Library (January 2015–October 2025) and citation chasing. Recommendation boundaries were anchored to AASM/ACP/European guidance. PRISMA-style exhaustive inclusion was not performed. Evidence was stratified by source: (A) direct perioperative clinical evidence; (B) extrapolated evidence; (C) mechanistic/consensus evidence.
Results: Perioperative sleep disturbances (PSD) are associated with postoperative pain, delirium/cognitive outcomes, and long-term reductions in quality of life. Perioperative RCTs/meta-analyses suggest that dexmedetomidine (DEX) and acupoint-related interventions may improve sleep and pain; however, optimal dose, timing, target populations, and monitoring strategies remain uncertain. Cognitive behavioral therapy for insomnia (CBT-I) is first-line treatment for insomnia (perioperatively, evidence is largely feasibility/pilot in nature), while digital CBT-I and wearable/bedside monitoring provide emerging tools for longitudinal follow-up and quality improvement (QI). Most studies have not stratified by phenotype; therefore, “phenotype–intervention” matching is supported mainly by mixed evidence, requiring explicit evidence labeling and restrained causal language.
Conclusion: A phenotype-guided pathway prototype may translate PSD risk profiling into combinable intervention modules (CBT-I/dCBT-I, DEX, acupoint/TEAS/electroacupuncture, analgesia and circadian management), while treating CSDS/CST-DS as a modulating axis. Prospective cohorts and phenotype-stratified RCTs are needed to validate the incremental value of the six phenotypes and module combinations, and to evaluate implementation models for digital interventions and remote monitoring.
Keywords
Perioperative sleep disturbances, Phenotype stratification, Mechanistic axes, Pathway prototype, Cognitive behavioral therapy for insomnia, Quality improvement
Introduction
Perioperative sleep disturbances (perioperative sleep disturbance, PSD) are common in surgical populations and are associated with clinically important outcomes, including suboptimal postoperative pain control, delirium, infection, cardiovascular events, and longer-term reductions in quality of life [1-5]. Prior studies have extensively described risk factors (psychological stress, surgical/anesthetic stress responses, inpatient environment, pre-existing sleep problems and comorbidities), yet a persistent clinical challenge remains: how to convert a “risk-factor checklist” into an executable assessment and intervention workflow within the time constraints of preoperative clinics and inpatient wards [1-5].
As a continuation of Part I, we proposed a five-axis mechanistic model and six candidate composite phenotypes for PSD, and positioned CSDS and the cervico-scapulo-thoracic-diaphragmatic-sympathetic (CST-DS) axis as a modulating/amplifying axis-useful for explaining additional load in specific populations, but not a prerequisite for executing the pathway [6]. The added value of Part II is operationalization: organizing tools and intervention components by “time window × phenotype × modules,” integrating perioperative evidence, and providing implementation interfaces that can be assembled locally and tracked through quality improvement (QI) [7,8]. Importantly, this paper proposes a conceptual prototype for hypothesis generation and prospective testing/local adaptation; it is not a validated clinical algorithm or decision tree.
This pathway is aligned with major guidelines: insomnia management follows the core principles of AASM/ACP and European consensus statements, prioritizing non-pharmacologic strategies with pharmacotherapy reserved for short-term, adjunctive use; Part II further specifies perioperative execution details for “risk stratification-exposure control-closed-loop follow-up” [9-12]. To mitigate concerns about salami slicing or duplicate publication, Part I focuses on mechanistic/phenotype conceptualization and falsifiable predictions, whereas Part II translates the framework into an implementable pathway prototype and monitoring architecture; the two parts are complementary with deliberate minimization of overlap [6-8].
Part II addresses three practical questions: (1) when time is limited, which minimal toolsets best support phenotype pre-screening? (2) how should interventions such as DEX, acupoint/electroacupuncture stimulation, CBT-I, and medications be organized into combinable modules by mechanistic axes and phenotypes, with evidence-tier constraints-(A) perioperative clinical evidence, (B) extrapolated evidence, (C) mechanistic/consensus-applied to both wording and boundaries of use? [13-16] (3) how can digital follow-up and QI methods embed PSD management into preoperative assessment, postoperative follow-up, and institution-level dashboards? [17] For CST-DS-related posture/breathing training, we designate this as an optional, C-level module primarily for patients with suspected cervicogenic load or high posture–ventilation burden; its incremental benefit requires prospective validation [7,8].
Search strategy and evidence selection
The objective of this paper is to propose an actionable pathway prototype and generate testable hypotheses, rather than to conduct an exhaustive systematic review. Given practical constraints, we adopted a purpose-driven, evidence-focused narrative synthesis. We prioritized major insomnia guidelines and high-quality systematic reviews/meta-analyses as the evidentiary backbone, then supplemented representative perioperative RCTs and prospective cohorts around key pathway nodes (preoperative screening, intraoperative exposure control, postoperative follow-up and modular interventions). Citation chasing was used to ensure transparent provenance for pivotal claims [18,19].
Database searches covered PubMed/MEDLINE, Embase, and the Cochrane Library (January 2015–October 2025). Search concepts combined “perioperative/postoperative” with sleep-related terms (sleep/insomnia/sleep disturbance/circadian), key modules (DEX, CBT-I/dCBT-I, digital CBT, acupuncture/electroacupuncture, TEAS, melatonin), and perioperative outcomes (sleep quality, arousal, delirium, pain, fatigue, quality of life). For minimal assessments, OSA/STOP-Bang and COMISA-related terms were added as needed [20-24]. Evidence was prioritized in descending order as systematic reviews/meta-analyses, RCTs, prospective cohorts, and high-quality real-world studies; mechanistic/animal studies were used only to support biological plausibility. To avoid over-inference in “phenotype-intervention” matching, evidence was labeled as (A) direct perioperative clinical evidence, (B) extrapolated evidence, or (C) mechanistic/consensus, with corresponding restraint in causal language. Because the paper emphasizes operationalization and hypothesis generation, PRISMA-style exhaustive inclusion/exclusion and quantitative pooling were not performed; subsequent prospective studies and phenotype-stratified validation trials are required to test and refine key hypotheses [19,20].
Time Windows and Tools: An Assessment Framework for Perioperative PSD
Here we operationalize the pathway as “time window × phenotype × intervention modules.” Abbreviations: TEAS, transcutaneous electrical acupoint stimulation; DEX, dexmedetomidine; COMISA, comorbid insomnia and sleep apnea. It should be emphasized that all pathway elements presented in Part II are prototypes. Where phenotype-specific direct evidence is unavailable, we apply qualified language and label evidence tiers consistently: (A) perioperative clinical evidence; (B) extrapolated evidence; (C) mechanistic/consensus. This paper does not provide a fixed decision tree that replaces guidelines; any “pathway recommendation” should be considered for routine implementation only after prospective validation [9-12].
Preoperative phase: Risk stratification and initial phenotype screening
The preoperative period is a key window for identifying high-risk PSD and establishing the foundation for intervention [1-3]. Given limited clinic time, a resource-tiered “minimal assessment package” is recommended. A core screen may use the ISI (2-3 minutes) plus brief pain/anxiety assessment (e.g., NRS and short-form measures) to rapidly identify high-arousal-pain coupling and high-burden patients [21]. An enhanced screen, for high-risk patients or pathway-based subspecialty programs, may add PSQI, ESS, and a brief CST-DS bedside posture screen (2-4 minutes), documenting neck/shoulder pain history, head/neck posture (forward head/rounded shoulders), scapular position, and thoracic mobility. Associated features (headache, dizziness, suboccipital tightness, upper-limb numbness) may suggest higher susceptibility driven by “posture–ventilation–cervicogenic load” [20,22]. For suspected OSA or multimorbidity, STOP-Bang and simplified oxygenation monitoring may be added to characterize complex COMISA/multiaxis overlap risk [23,24].
Intraoperative phase: Sedation-analgesia structure and CST-DS axis protection
Intraoperative management focuses on three modifiable exposure domains: depth/structure of sedation-analgesia and opioid burden, hemodynamic stability, and positioning/airway management [1,4]. Perioperative systematic reviews and RCTs suggest that DEX may improve postoperative sleep and pain, but effects vary by dose, timing, and patient heterogeneity. Accordingly, DEX may be better conceptualized as an adjunct for tuning autonomic-endocrine and drug-network axes rather than as a stand-alone sedative [13]. Use should be co-designed with opioid-sparing multimodal analgesia and COMISA risk, with titration under appropriate monitoring [13,24]. For patients with cervical degeneration or prior cervical surgery, positioning and fixation should avoid prolonged extreme flexion/extension, favoring neutral alignment (or mild flexion where appropriate), supported by imaging/monitoring when indicated, and preserving an interface for postoperative “safe posture” training [7,8].
Postoperative phase: Symptom monitoring and digital phenotyping
Postoperative nights 3-5 are often a PSD peak and represent a critical window to assess response and dynamically adjust modules [1-4]. In high-risk or research settings, PSG or simplified PSG can quantify arousal density, sleep staging, and respiratory events; in general wards, actigraphy, bedside sensors, or continuous oximetry can be paired with questionnaires to construct a “symptoms × activity × oxygenation” digital phenotype [5,17]. Existing studies suggest that sleep duration, arousal/rest-activity rhythms, and related metrics correlate with cognitive outcomes, inflammatory markers, and fatigue, supporting sleep as a candidate biomarker for perioperative risk prediction and rehabilitation planning. To emphasize sleep as a quantifiable entry point for risk/recovery phenotyping, we propose a “symptoms × activity × oxygenation” prototype (see Figure 1) to align routine monitoring with actionable stratified management.
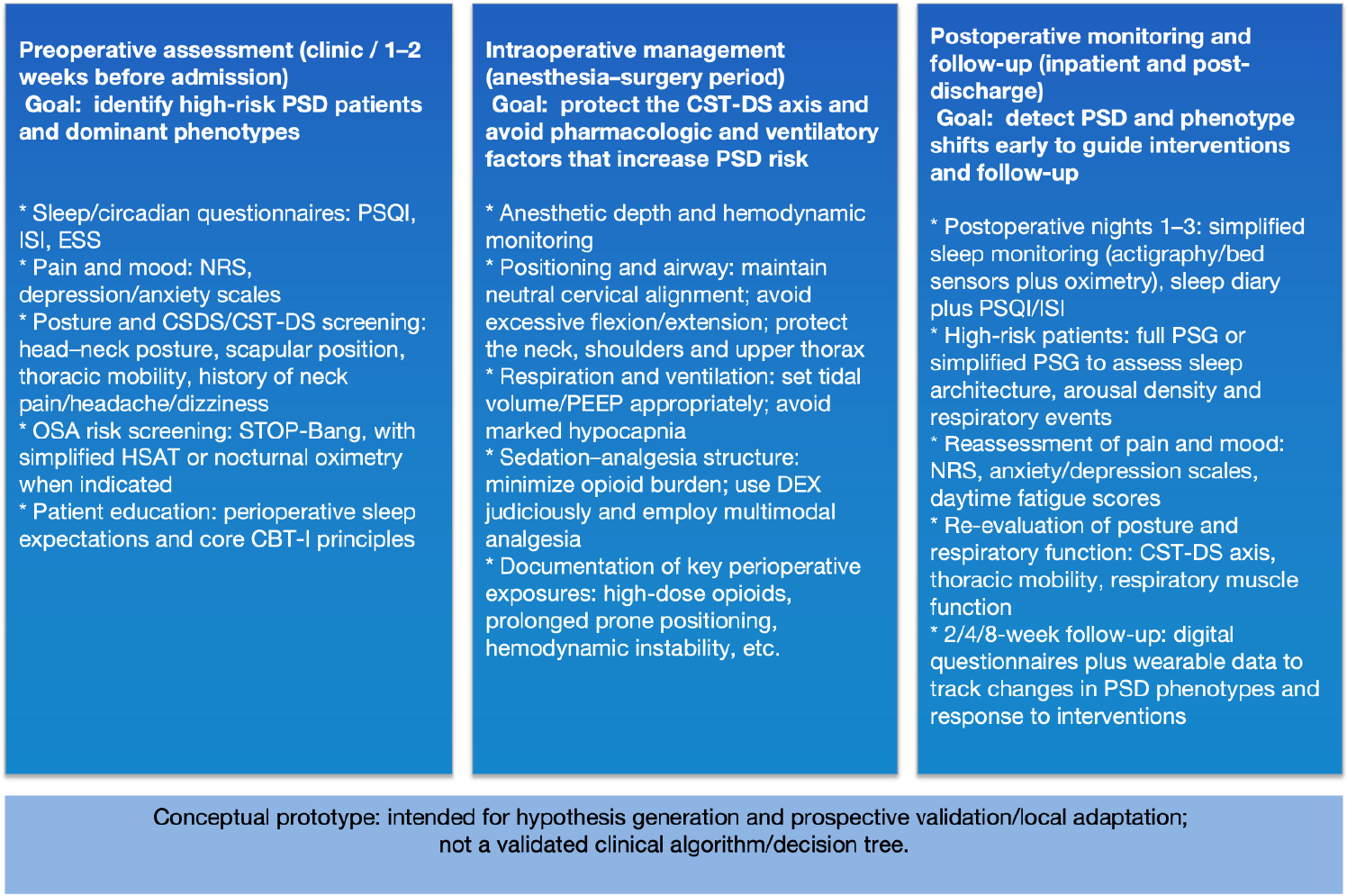
Figure 1: Postoperative “symptom × activity × oxygenation” digital phenotyping prototype.
Legend: This figure outlines a pragmatic, perioperative data-capture prototype to support postoperative sleep disturbance (PSD) risk stratification and early phenotype tracking. The preoperative domain focuses on identifying high-risk patients and dominant phenotypes using brief sleep/circadian questionnaires, pain-mood screening, posture/CST-DS screening, and OSA risk assessment. The intraoperative domain highlights exposure control and CST-DS axis protection (neutral cervical alignment, ventilatory parameter optimization, and minimization of high-dose systemic opioids). The postoperative domain emphasizes early detection of PSD and phenotype shifts (nights 1-3 as a simplified monitoring window when feasible), reassessment of pain/mood and daytime fatigue, and longitudinal follow-up (e.g., 2/4/8 weeks) using questionnaires and optional wearable data. This is a conceptual prototype intended for hypothesis generation and prospective validation/local adaptation; it is not a validated clinical algorithm or decision tree.
Abbreviations: CBT-I: Cognitive Behavioral Therapy for Insomnia; CST-DS: Cervico-Scapulo-Thoracic-Diaphragmatic-Sympathetic Axis; DEX: Dexmedetomidine; ESS: Epworth Sleepiness Scale; HSAT: Home Sleep Apnea Test; ISI: Insomnia Severity Index; NRS: Numeric Rating Scale; OSA: Obstructive Sleep Apnea; PEEP: Positive End-Expiratory Pressure; PSG: Polysomnography; PSD: Perioperative Sleep Disturbance; PSQI: Pittsburgh Sleep Quality Index
Feasibility and resource tiering
To support implementation across institutions, we recommend explicitly defining a three-tier model-core, enhanced, and advanced. In low-resource settings, the emphasis is on minimal screening, environmental/circadian management, multimodal analgesia, and simplified monitoring. In medium-resource settings, these can be supplemented with embed-able CBT-I elements, posture/positioning screens, and accessible modules such as TEAS. In high-resource or research environments, dCBT-I, continuous wearable/bedside monitoring, and QI dashboards can be deployed. Across tiers, local SOPs should clearly distinguish “core” from “optional” elements and use PDSA cycles for iterative scaling rather than one-time full deployment [9-12].
To facilitate rapid “phenotype-module” matching across resource settings, we summarize each phenotype’s dominant mechanistic axes, minimal assessment package, and A/B/C evidence tiering in Table 1 Table 1 addresses “when to assess” and “what to assess,” but clinical execution also requires aligning time windows with phenotype–module matching into an actionable pathway. We therefore propose the “time window × phenotype” workflow prototype in Figure 2, designed to align preoperative screening, intraoperative exposure control, and postoperative monitoring/follow-up into a closed loop. Importantly, the figure is a conceptual prototype intended for communication and hypothesis generation; phenotype-specific incremental benefit requires prospective phenotype-stratified validation.
Table 1: Prototype mapping between PSD phenotypes and intervention modules (distinguishing evidence level A vs B/C).
|
PSD phenotype |
Likely dominant mechanistic axes |
Core assessment (clinic/ward) |
Perioperative evidence-supported modules (A) |
Adjunctive / extrapolated modules (B/C) |
Key safety / feasibility notes |
|
Type 1: Psychological-nociceptive high-arousal dominant phenotype |
Axes 3-4 ± Axis 5 |
ISI / brief sleep items [21]; pain NRS; rapid mood screening |
Multimodal analgesia to reduce systemic opioid exposure (principle-level evidence) [1,3,4] |
Abbreviated CBT-I modules [9,15]; dCBT-I [17]; relaxation training; environmental optimization [1,3] |
Avoid causal language; implement with resource-tiering and local feasibility constraints [1,3] |
|
Type 2: Drug-anesthesia-respiratory-control dominant phenotype |
Axis 4-5 ± OSA/COMISA |
OSA screening (STOP-Bang) [23]; oximetry when indicated/feasible; medication review |
Reduce systemic opioids; DEX as an adjunct in selected patients (titrated) [13] |
Sleep hygiene / environmental optimization [1,3]; referral to sleep medicine when needed (prototype) |
Maintain high vigilance for hypoxemia; avoid routine postoperative benzodiazepine hypnotics (guideline-oriented) [11] |
|
Type 3: Inflammation-immune-glymphatic vulnerability phenotype |
Axis 2 ± Axis 3 |
Signals of inflammatory/infectious risk; fatigue/recovery scales when feasible; sleep-quality tracking [5] |
Multimodal analgesia; acupoint-related interventions have perioperative evidence in selected procedures [14] |
CBT-I as an adjunct [9,15]; nutrition/activity optimization; biomarker tracking (prototype) [5] |
High heterogeneity-avoid over-claiming “anti-inflammatory causal effects” [5] |
|
Type 4: CST-DS / cervicogenic dominant phenotype |
Axis 1 ± Axis 3 |
Brief posture/neck screening; position-dependent symptoms; snoring differences across sleep positions (prototype) [6-8] |
Position protection; posture/breathing guidance with rehabilitation-team involvement (prototype) [6-8] |
TEAS/acupressure [14]; CBT-I adjunct [9]; activity logging and posture tracking (prototype) |
Rule out cervical instability/high-risk pathology first; prioritize safe positioning [6-8] |
|
Type 5: Environment-process-circadian dysregulation phenotype |
Axis 5 ± Axis 3 |
Ward/environment audit; sleep phase/timing; light/noise exposure [1,3] |
Noise/light control; nursing bundles; early mobilization (review- and pathway-level evidence) [1,3] |
Melatonin for circadian realignment (evidence largely from aggregated studies) [16]; dCBT-I follow-up [17]; rhythm tracking (prototype) |
Make “core vs optional” explicit; staffing and workflow constraints are central determinants [1,3] |
|
Type 6: Complex COMISA / multi-axis overlap phenotype |
Multiple axes |
Comprehensive assessment as above; prioritize identification of the highest-risk signals [2,24] |
Combined intervention bundles based on risk and monitoring results (prototype) [2,24] |
dCBT-I plus monitoring [17]; phenotype re-assessment over time (prototype) |
Start simple, then escalate-avoid “one-size-fits-all bundles”; refer and collaborate when needed [24] |
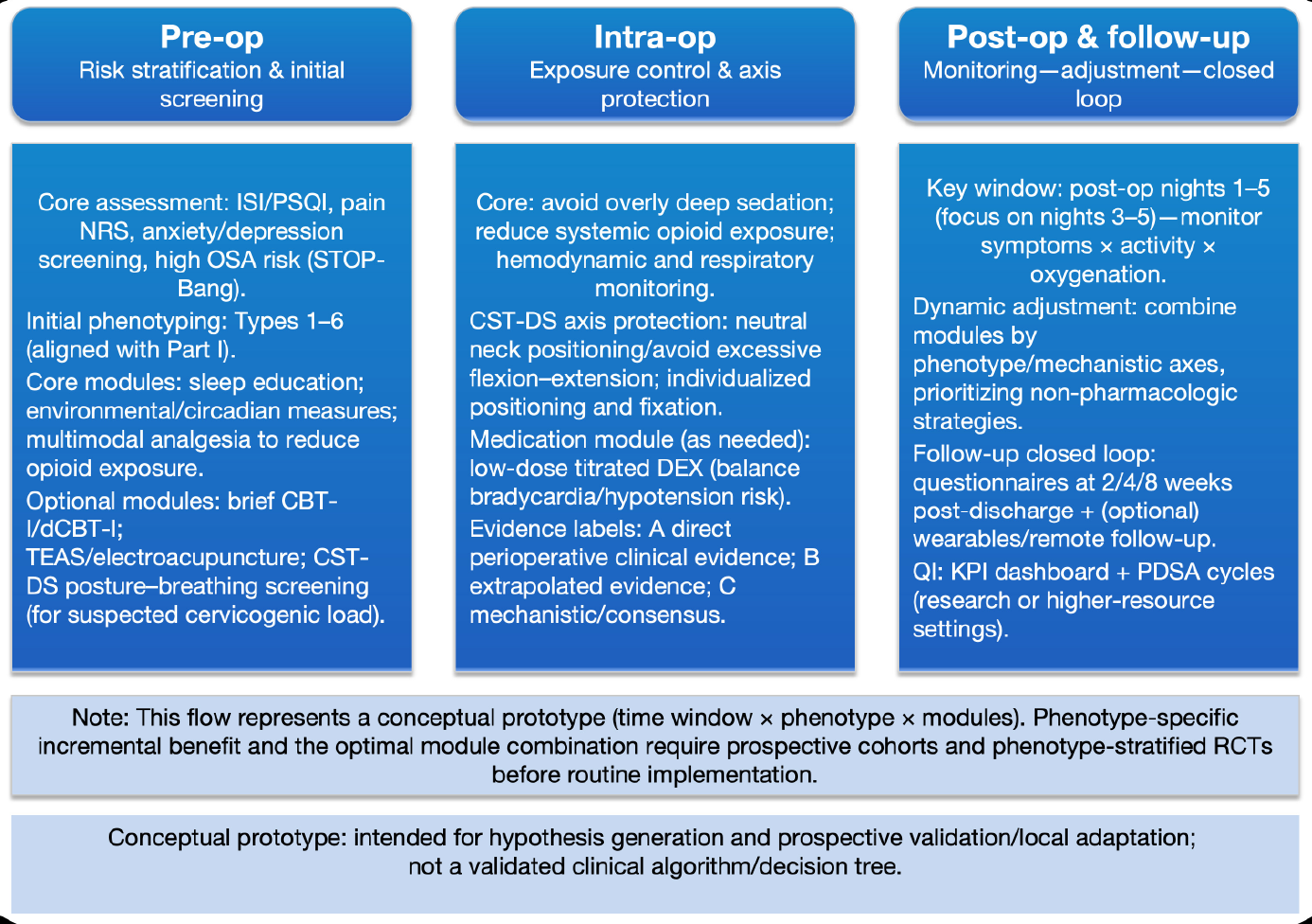
Figure 2: Time-window × phenotype pathway flow for perioperative PSD management (conceptual).
Legend: This flow diagram operationalizes PSD care as an iterative pathway driven by time windows (pre-op, intra-op, post-op/follow-up), phenotype labels (Types 1-6; aligned with Part I), and modular interventions. Preoperatively, a core screening set (ISI/PSQI, pain NRS, anxiety/depression screening, OSA risk) supports initial phenotyping and selection of core modules (sleep education, environmental/circadian measures, multimodal analgesia to reduce opioid exposure), with optional modules (brief CBT-I/dCBT-I, TEAS/electroacupuncture, CST-DS posture–breathing screening) when indicated. Intraoperatively, the pathway emphasizes exposure control and axis protection (avoid overly deep sedation, reduce systemic opioid exposure, monitor hemodynamics and respiration; protect cervical alignment), with judicious medication modules (e.g., low-dose titrated DEX) based on risk–benefit. Postoperatively, the key monitoring window is nights 1-5 (with emphasis on nights 3-5 where appropriate), and modules are dynamically combined to prioritize non-pharmacologic strategies and close the follow-up loop (questionnaires at 2/4/8 weeks; optional wearable/remote follow-up). Evidence labels (A/B/C) denote direct perioperative clinical evidence, extrapolated evidence, and mechanistic/consensus support, respectively. The pathway is a conceptual prototype; phenotype-specific incremental benefits and optimal module combinations require prospective cohorts and phenotype-stratified RCTs before routine implementation.
Abbreviations: CBT-I: Cognitive Behavioral Therapy for Insomnia; CST-DS: Cervico-Scapulo-Thoracic-Diaphragmatic-Sympathetic axis; dCBT-I: Digital CBT-I; DEX: Dexmedetomidine; ISI: Insomnia Severity Index; KPI: Key performance indicator; NRS: Numeric Rating Scale; OSA: Obstructive Sleep Apnea; PDSA: Plan-Do-Study-Act; PSD: Perioperative Sleep Disturbance; PSQI: Pittsburgh Sleep Quality Index; RCT: Randomized Controlled Trial; TEAS: Transcutaneous Electrical Acupoint Stimulation
Phenotype-Guided Nonpharmacologic Interventions: From CBT-I to a CST-DS-Informed Postural Pathway
CBT-I and structured sleep education
Systematic reviews and major guidelines consistently recommend cognitive behavioral therapy for insomnia (CBT-I) as first-line treatment for chronic insomnia in adults. CBT-I reduces insomnia severity, improves sleep efficiency, and shortens sleep-onset latency; it may also benefit co-occurring symptoms such as anxiety and depression [9-12,15,25]. When delivering a full CBT-I program is not feasible in the perioperative setting, CBT-I can be “unbundled” into lightweight modules: brief preoperative education and core strategies 1-2 weeks before surgery (stimulus control, simplified sleep restriction, and reducing catastrophizing), followed by postoperative reinforcement and re-assessment through follow-up visits or digital formats [9,10,12,25].
Evidence tier: CBT-I/dCBT-I: B. As a cornerstone nonpharmacologic module, perioperative outcome benefits still require prospective validation [9,10,12,17].
CST-DS-informed postural and cervical interventions
For patients with suspected high “posture-ventilation-cervicogenic load,” this prototype treats CST-DS measures as an optional module: included when screening suggests relevant load and omitted when implementation is not feasible. Preoperatively, head–neck posture, scapular position, thoracic mobility, and cervical range of motion are documented, with imaging used when indicated. Postoperatively, early retraining focuses on “neutral cervical alignment, scapular stability, thoracic expansion, and diaphragmatic engagement,” integrated with sleep-position counseling and pillow-height guidance [6-8].
Safety: Avoid or defer such training in patients with cervical instability, acute trauma, progressive neurologic deficits, or severe myelopathy.
Evidence tier: CST-DS / posture-breathing: C [7,8].
Acupoint Stimulation, Electroacupuncture, and TEAS
Perioperative RCTs and systematic reviews suggest that transcutaneous electrical acupoint stimulation (TEAS), acupressure, and electroacupuncture may improve subjective postoperative sleep-related measures in some surgical populations, alongside analgesic effects, reduced PONV, and lower opioid requirements. However, substantial heterogeneity in stimulation parameters, acupoint selection, and patient populations remains, and should be addressed in phenotype-stratified studies [14,26,27]. Mechanistic studies provide biologic plausibility through inflammatory signaling, autonomic modulation, and nociceptive regulation, but claims should not be framed as causal “anti-inflammatory” or “immune-remodeling” effects without direct perioperative clinical confirmation [5,28].
Safety: Use electrical stimulation cautiously in patients with pacemakers/ICDs; avoid compromised or infected skin. For acupuncture/electroacupuncture, assess coagulopathy/anticoagulation and infection risk, and follow credentialing and procedural standards.
Evidence tier: TEAS/electroacupuncture: A-B. Unless direct perioperative evidence is available, results are described as “suggestive” or “may confer benefit” [14,26,27].
To reduce interdisciplinary communication burden and support bedside decision-making, we visualize the “phenotype-module” mapping as a matrix (see Figure 3). The matrix explicitly distinguishes core versus optional modules, enabling a minimum viable pathway across resource settings.
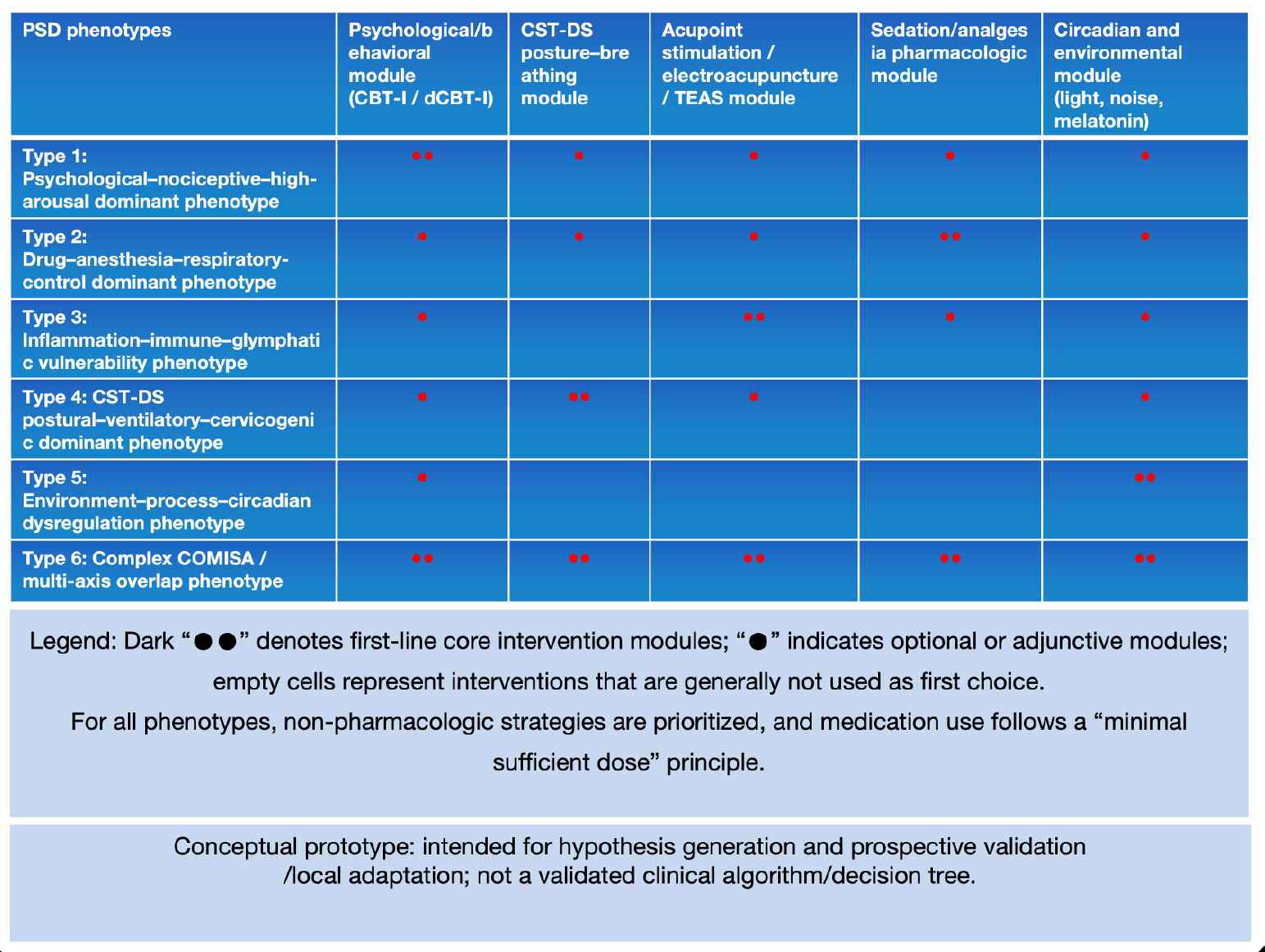
Figure 3: Phenotype-module matrix visualization for PSD management (conceptual).
Legend: This matrix visualizes the recommended alignment between six PSD phenotypes (Types 1-6) and candidate intervention modules. Dark markers (“●●”) denote first-line core modules for a given phenotype, whereas single markers (“●”) indicate optional or adjunctive modules; blank cells represent interventions that are generally not used as first choice. Across phenotypes, non-pharmacologic strategies are prioritized, and medication use should follow a guideline-consistent “minimal sufficient dose” principle and be used short-term/as adjuncts. The matrix is intended as an implementation-oriented prototype to support structured tailoring and future falsifiable testing, rather than as a prescriptive decision algorithm.
Abbreviations: CBT-I: Cognitive Behavioral Therapy for Insomnia; COMISA: Comorbid Insomnia and Sleep Apnea; CST-DS: Cervico-Scapulo-Thoracic-Diaphragmatic-Sympathetic Axis; dCBT-I: Digital CBT-I; DEX: Dexmedetomidine; PSD: Perioperative Sleep Disturbance; TEAS: Transcutaneous Electrical Acupoint Stimulation
Vignette: Type 4
A patient presents with difficulty initiating sleep preoperatively and morning neck stiffness; STOP-Bang indicates moderate-to-high risk, and posture screening shows forward head posture with restricted thoracic expansion [23,24]. The pathway prototype proceeds as follows:
Preoperative (1-2 weeks): Brief education plus simplified CBT-I, combined with gentle posture-breathing retraining;
Intraoperative: Maintain neutral cervical positioning, avoid excessive flexion/extension or prolonged pressure, use multimodal analgesia to reduce opioids, and titrate DEX when hemodynamically appropriate;
Postoperative nights 1-3: Simplified oxygenation/activity monitoring plus a sleep diary, with reassessment of pain and fatigue;
After discharge (weeks 2/4/8): Electronic scales and/or wearables for follow-up, with escalation to dCBT-I and intensified rehabilitation if needed [7,8,13,17]. This vignette is intended to illustrate implementation of the prototype; phenotype-specific incremental effects remain to be tested in stratified studies [1-5].
Pharmacologic and Anesthetic Strategies: “Minimum Effective Dosing” Through a Mechanistic-Axis Lens
Nonpharmacologic measures are foundational to PSD management, yet perioperative practice cannot always avoid sedative/hypnotic exposure. The central goal-consistent with AASM/ACP and European guidance-is to prioritize nonpharmacologic approaches while using medications as short-term, adjunctive therapy at the lowest effective dose, avoiding unnecessary disruption of sleep architecture or exacerbation of respiratory events [9-12]. In older adults, patients at high OSA risk, and those at elevated delirium risk, sedative burden should be minimized in alignment with institutional policy and guideline principles [4,9-12,24].
Opioids and benzodiazepines can suppress respiratory drive, reduce upper-airway muscle tone, and alter sleep architecture, potentially worsening OSA/CSA and increasing risks of hypoventilation and delirium. Opioids may also reduce slow-wave sleep and REM sleep while increasing nocturnal awakenings [4,24,29]. Accordingly, for phenotypes dominated by “drug-anesthesia-respiratory control” mechanisms (Type 2), opioid-sparing multimodal analgesia is prioritized, and benzodiazepines are generally avoided as routine postoperative hypnotics [9-12,24,29].
Dexmedetomidine (DEX), supported by perioperative meta-analyses and selected RCTs, has shown signals of improved subjective sleep outcomes and analgesia-related measures. However, effects vary with dose, timing, and patient characteristics; and in some critically ill populations, objective sleep improvements have been inconsistent. DEX is therefore best positioned as a context-dependent adjunct that requires careful titration, balancing potential benefit against bradycardia and hypotension risk [13,16,30-32].
DEX as an adjunct sedation-analgesia option in selected patients
DEX may be particularly appropriate for patients with prominent sympathetic activation and marked pain and/or anxiety, and for phenotypes where tuning the “autonomic-endocrine axis” and “drug-neural network axis” is prioritized. Clinicians should explicitly weigh potential sleep/analgesia benefits against risks of bradycardia and hypotension, and avoid routine use in patients with severe baseline bradycardia, advanced conduction block without pacing, or hemodynamic instability [13,13,30-32].
Melatonin and other hypnotics
Melatonin and melatonin-receptor agonists may facilitate circadian realignment and sleep initiation, with relatively limited impact on respiratory drive [16]. In older hospitalized and perioperative-related studies, systematic reviews/meta-analyses suggest a possible reduction in delirium risk, although evidence quality and heterogeneity warrant cautious interpretation [33]. When CBT-I cannot be implemented in the short term or remains incomplete, guidelines recommend short courses-after assessing risks such as falls, cognitive impairment, and dependence-of non-benzodiazepine receptor agonists, melatonin-receptor agonists, or low-dose doxepin as alternative/adjunctive options [10-12]. In perioperative care, melatonin is best paired with light/noise/schedule management; traditional sedative-hypnotics should be minimized, and ideally avoided in patients at high OSA risk or with limited respiratory reserve [10-12,24].
Evidence tier: melatonin/circadian agents: B [16,33].
To support scalability and patient safety, we summarize applicability and boundary conditions (Table 2) to clarify when disease-specific guidelines or specialty pathways should take precedence.
Table 2: Prototype perioperative PSD management: Safety boundaries and alignment points with local SOPs.
|
Module |
Main risks |
Contraindications / cautions |
Monitoring and mitigation (prototype) |
|
Bradycardia, hypotension; oversedation (especially with other sedatives) |
High-grade AV block/severe sinus bradycardia; hemodynamic instability; use cautiously in severe heart failure or significant valvular disease |
Start low and titrate; continuous BP/HR monitoring; reduce opioids/other sedatives synergistically; discontinue and manage as needed (fluids, vasopressors, atropine, etc.) |
|
|
TEAS / electroacupuncture / acupoint stimulation [evidence: [14,26,27]] |
Skin irritation; bleeding/hematoma; infection; vasovagal syncope (rare) |
Coagulopathy or therapeutic anticoagulation with high puncture risk; local skin infection/ulceration; implanted pacemaker/ICD (requires assessment and risk mitigation for electrical stimulation) |
Strict asepsis and contraindication screening; use safe current intensity and electrode placement; document adverse events; align with institutional credentialing and procedural workflows |
|
CST-DS posture/neck interventions and breathing training [evidence: [7,8]] |
May exacerbate cervical instability/neurologic symptoms; pain-triggered muscle spasm |
Avoid if suspected cervical instability, spinal cord compression, or progressive neurologic deficit; restrict activity after recent cervical surgery per surgical guidance |
Screen for red flags first; emphasize gentle training; consult spine surgery/rehabilitation when needed; escalate stepwise with symptoms as boundaries |
|
Sedative-hypnotics (benzodiazepines and non-benzodiazepines) [evidence: [10-12,24]] |
Falls, delirium, respiratory depression; dependence/rebound insomnia |
High-risk OSA or limited respiratory reserve; older adults/cognitive impairment; high delirium risk; concurrent opioids/other sedatives |
Prioritize non-pharmacologic strategies; if needed: shortest duration and lowest effective dose; enhance overnight monitoring and fall prevention; guideline-consistent prescribing |
|
Melatonin / circadian module [evidence: [16,33]; guidelines: [10-12]] |
Somnolence (usually mild); potential drug interactions |
Individualized assessment (hepatic function/interactions); not a substitute for monitoring and treating respiratory events |
Combine with light/noise and schedule management; define as an adjunct module; record sleep response and adverse events; use short-term and at the lowest effective dose |
Note: The above list summarizes safety-boundary prompts for a “pathway prototype”. Implementation must be aligned with institutional policies, credentialing, and local SOPs, and should be updated iteratively during prospective deployment. Evidence level statement (prototype): for DEX, evidence is primarily A (perioperative RCTs/meta-analyses), suggesting potential benefit for subjective sleep and some objective metrics; however, phenotype-stratified, phenotype-specific incremental benefit remains insufficient. This prototype does not position DEX as a universal hypnotic substitute; it emphasizes context dependence, low-dose titration, and explicit safety boundaries.
Digital Phenotyping and Quality Improvement: From Individual Cases to Dashboards
Making PSD management operational at scale requires more than individualized case handling; it requires a longitudinal metric system that clarifies the burden of PSD, intervention coverage, and outcome trajectories. [1-5,7,8] Digital phenotyping and remote follow-up are optional tools: implementation depends on local resources, data governance, and privacy/compliance infrastructure. A phased rollout (pilot first, then expansion) is generally preferable to institution-wide deployment at the outset [34-36].
Digital CBT-I (dCBT-I)
dCBT-I has shown overall effectiveness comparable to in-person CBT-I across chronic insomnia and multiple comorbidity contexts, while offering advantages in scalability and closed-loop follow-up [23,25,26,30,33]. In perioperative care, dCBT-I can be used to “shift earlier and extend longer”: short-course preoperative preparation for at-risk patients, followed by postoperative reassessment and reinforcement at weeks 2, 4, and 8. This supports linkage of sleep, pain, and mood trajectories over time and can improve adherence through reminders to sleep prescriptions and posture/breathing exercises [1-3,5,8,30,33,34].
Wearables and bedside monitoring: Tracking trajectories rather than a single night
Actigraphy, bedside sensors, and continuous oximetry/heart-rate monitoring can capture nighttime activity, HRV, and oxygenation patterns with relatively low burden. Combined with questionnaire measures, these signals support multidimensional digital phenotyping and help identify which mechanistic axis is most likely driving the problem [5,7,8,15,34]. Within the multiaxis framework, monitoring can be simplified as follows: Type 1 emphasizes arousal density and nocturnal heart rate; Type 2 emphasizes hypoxemia burden and respiratory events; Type 3 pairs inflammatory markers with changes in sleep efficiency; Type 4 focuses on snoring/oxygenation under different sleep positions; Type 5 evaluates re-establishment of rest–activity rhythms; and Type 6 integrates multiple signals [5,7,8,15,24,34].
KPI dashboards and PDSA cycles
Recommended dashboard indicators include: preoperative high-risk identification rate, proportion of moderate-to-severe PSD within 7 postoperative days, coverage of stratified interventions (CBT-I/acupoint-related modules/DEX, etc.), readmission rates and long-term fatigue/quality-of-life outcomes, and rates of key complications such as severe desaturation and delirium [1-5,7,8,15,16,22,23,30]. To move from “single-case experience” to auditable team-based improvement, we propose a phenotype-stratified KPI dashboard and PDSA closed loop (see Figure 4). The dashboard is intended to track coverage, outcomes, and escalation-path consistency, enabling periodic review and local iteration.
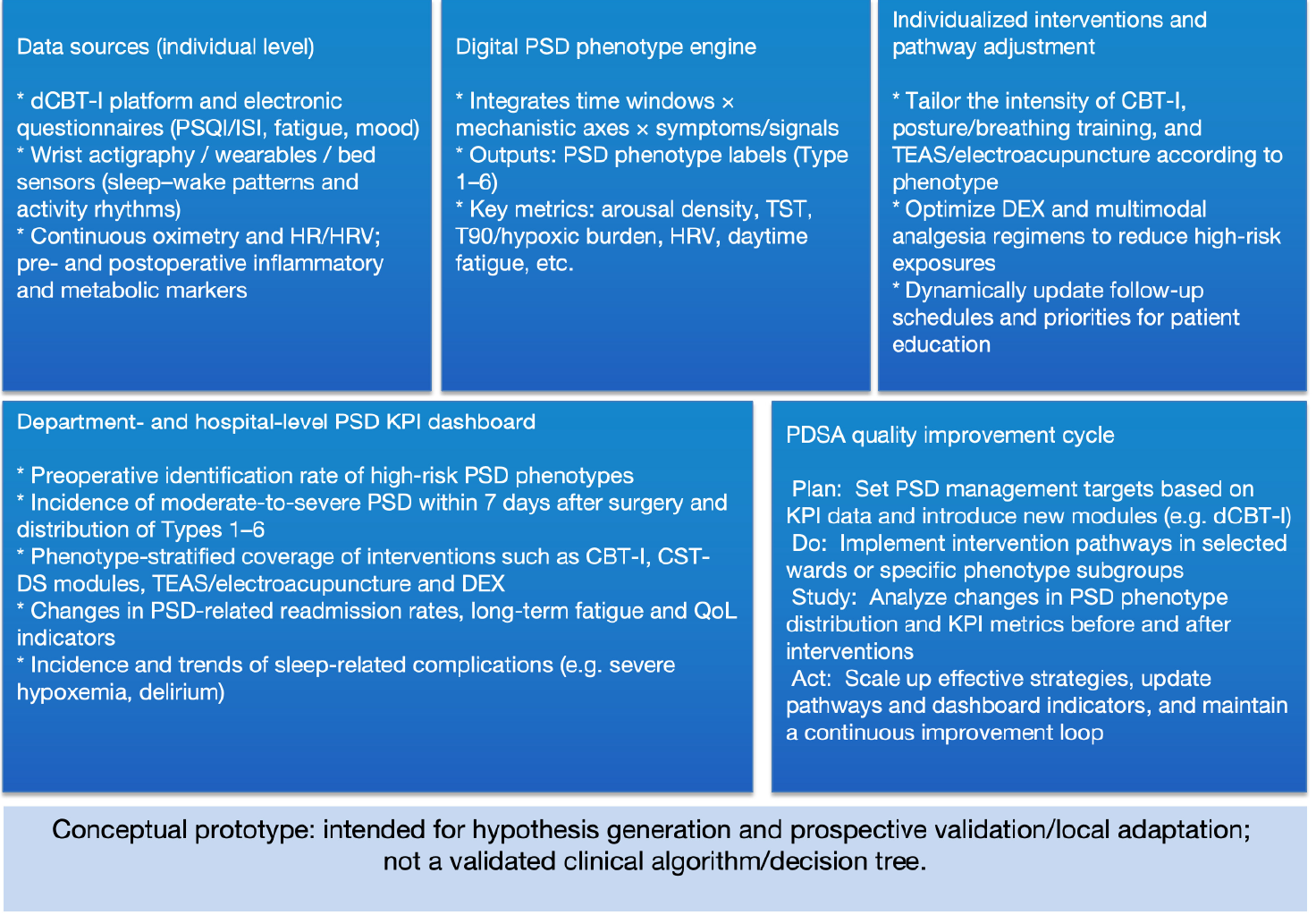
Figure 4: PSD KPI dashboard and PDSA quality-improvement loop (conceptual).
Legend: This figure depicts a program-level implementation architecture that links individual-level data sources (questionnaires, actigraphy/wearables, continuous oximetry, HR/HRV, and selected biomarkers) to a digital PSD phenotype engine (time windows × mechanistic axes × symptoms/signals) and phenotype-guided pathway adjustment. At the department/hospital level, a PSD KPI dashboard can track screening coverage, PSD incidence/severity, phenotype distributions, intervention coverage, and longer-term outcomes (e.g., readmissions, fatigue and quality-of-life indicators), enabling iterative pathway refinement through PDSA cycles (plan targets, implement in selected settings, analyze KPI shifts, and scale/update). This is a conceptual prototype for hypothesis generation and prospective validation/local adaptation; it is not a validated clinical algorithm or decision tree.
Abbreviations: HR: Heart Rate; HRV: Heart Rate Variability; ISI: Insomnia Severity Index; KPI: Key Performance Indicator; PDSA: Plan-Do-Study-Act; PSD: Perioperative Sleep Disturbance; PSQI: Pittsburgh Sleep Quality Index; QoL: Quality of Life; T90: Time with Oxygen Saturation < 90%; TST: Total Sleep Time
Discussion
In Part II, we translate the “etiology-mechanism map” into a pathway prototype organized by “time window × phenotype × intervention modules.” It bears repeating: this paper presents a conceptual prototype for hypothesis generation, prospective testing, and local adaptation; it is not a validated algorithm or fixed decision tree, nor does it impose mandatory requirements for routine care.
The value of the prototype lies in aligning fragmented evidence with clinical touchpoints: preoperatively, using low-burden screening to identify high-risk patients and embedding lightweight CBT-I/sleep education; intraoperatively, limiting harmful exposures through “minimum effective” sedation–analgesia and positioning/airway management; postoperatively, using a symptom-monitoring-follow-up loop to support modular escalation/de-escalation and QI [1-5,9-13,15,16,22,23,30,33]. As a working hypothesis, combining DEX, acupoint-related stimulation, and multimodal analgesia by mechanistic axes may improve sleep and pain without increasing sedation burden or respiratory adverse events; dCBT-I and wearable/bedside monitoring provide practical interfaces for longitudinal follow-up and dashboard-based management [13,16-18,22,23,30,34-36].
Limitations should be stated upfront
First, most studies have not performed phenotype- or mechanistic-axis–stratified randomization or prespecified subgroup analyses, limiting inference about phenotype-specific benefit. Second, evidence quality varies across modules, and some linkages remain reliant on extrapolation or mechanistic reasoning. Third, CSDS/CST-DS-related postural and cervical measures are not yet standardized, and brief, reliable bedside scoring tools are lacking. Fourth, implementation feasibility depends heavily on resources and credentialing, and low-resource settings may be limited to core elements [15,7,8,13,16,22-24,33-36]. Additional context is also important: some evidence is disproportionately derived from Asian settings (particularly for TEAS/electroacupuncture), so cross-cultural generalizability requires caution; the authors’ research background may introduce emphasis bias, which is why this paper constrains conclusions using A/B/C evidence tiering and deliberately restrained language [13,22,33].
Future work can follow three pragmatic tracks: (1) prospective cohorts collecting multiaxis measures and digital signals to test whether phenotypes “naturally emerge” via unsupervised methods and how they relate to pain, delirium, long-term fatigue, and quality of life; (2) phenotype-stratified validation RCTs comparing combinations of CBT-I, CST-DS-informed rehabilitation, DEX strategies, and acupoint-related interventions, with both incremental benefit and safety endpoints; and (3) development of a shareable minimum dataset and interoperable data structure to enable multicenter QI and real-world research [34-36]. Overall, anchored to the core principles of AASM/ACP/European guidance, the prototype offers an executable, testable, and iteratively refinable blueprint for perioperative PSD management [9-12,33,34].
Conclusion
Perioperative sleep disturbances (PSD) represent an actionable risk factor that influences pain, delirium, hypoxemic events, and longer-term fatigue and quality-of-life outcomes. This work proposes a pathway prototype organized by “time window × phenotype × intervention modules”: preoperatively, low-burden tools support risk stratification and phenotype pre-screening, enabling early lightweight CBT-I/sleep education; intraoperatively, exposure control emphasizes opioid and oversedation reduction, positioning/airway management, and context-dependent DEX use when appropriate; postoperatively, nights 3-5 serve as a key window for constructing a “symptoms-activity-oxygenation” digital phenotype using feasible monitoring, supporting modular titration and a closed-loop follow-up strategy.
Crucially, this pathway is positioned as an actionable conceptual prototype for hypothesis generation, prospective testing, and local adaptation-not as a validated algorithm or mandatory decision tree. The major evidence gap is the lack of phenotype- or mechanistic-axis–stratified randomization and prespecified subgroup analyses in most studies, leaving phenotype-specific benefit uncertain. Intervention protocols, dosing, timing, and outcome measurements are also heterogeneous, and CST-DS-related postural/cervical indicators still lack standardized bedside scoring. Future progress requires: (1) prospective cohorts integrating multiaxis measures and digital signals to verify phenotype “natural emergence”; (2) phenotype-stratified validation RCTs testing incremental benefit from combinations of CBT-I, CST-DS-informed rehabilitation, DEX strategies, and acupoint-related interventions; and (3) shared minimum datasets and QI dashboards to move PSD management from case-by-case practice toward traceable, iterative precision care.
Author Contributions
QY: Conceptualization, Methodology, Drafting of the initial manuscript;
FFL: Literature synthesis, Drafting of the initial manuscript;
MYC: Figures/Tables and Visualization;
YES/JFW: Critical review and revision of key content;
WC: Conceptualization, Supervision, Funding acquisition, Final approval.
All authors approved the final version and agree to be accountable for all aspects of the work.
Conflicts of Interest
None.
Funding
National Natural Science Foundation of China (Project Approval Number: 81200858); Jiangsu Province 333 High-level Talent Training Project [Certificate No.: (2022) No. 3-10-007].
Clinical Trials from Nanjing Drum Tower Hospital, Affiliated Hospital of Medical School, Nanjing University, the Huai'an Matching Assistance Special Project (2024-2025.
AI Use Statement
• We used large language model-based tools (e.g., ChatGPT) solely for language editing, formatting support, and improving clarity.
• AI tools were not used to generate new scientific conclusions, fabricate evidence, or replace the authors’ scholarly judgment; all key ideas, structure, and final wording were reviewed and revised by the author team, which takes full responsibility for the manuscript.
• The authors assume full responsibility for the accuracy, originality, and citation integrity of all content.
References
- Lin D, Huang X, Sun Y, Wei C, Wu A. Perioperative sleep disorder: a review. Front Med (Lausanne). 2021;8:640416. doi:10.3389/fmed.2021.640416.
- Butris N, et al. Prevalence and risk factors of sleep disturbances among surgical patients: a systematic review and meta-analysis. Sleep Med Rev. 2023;69:101786. doi:10.1016/j.smrv.2023.101786.
- Sipilä RM, Kalso EA. Sleep well and recover faster with less pain—A narrative review on sleep in the perioperative period. J Clin Med. 2021;10(9):2000. doi:10.3390/jcm10092000.
- Chouchou F, et al. Postoperative sleep disruptions: a potential catalyst of postoperative pain? Sleep Med Rev. 2014;18(3):273–282. doi:10.1016/j.smrv.2013.07.002.
- Liu Y, Wu F, Zhang X, Jiang M, Zhang Y, Wang C, et al. Associations between perioperative sleep patterns and clinical outcomes in patients with intracranial tumors: a correlation study. Front Neurol. 2023;14:1242360. doi:10.3389/fneur.2023.1242360.
- Yin Q, Cheng MY, Wang S, Sun YE, Wang JF, Cheng W. Cervicogenic Sleep Disorder Syndrome: Definition and Classification Recommendations. Transl Perioper Pain Med. 2025;12(1):746–767. doi:10.31480/2330-4871/205.
- Yin Q, Zheng Y, Wang S, Sun YE, Wang JF, Cheng W. “Postural Alignment First, Symptom Tailored” Integrated Pathway as a Multimodal Peripheral Intervention Strategy for Pain and Cervical Disease (Part 1). Transl Perioper Pain Med. 2025;12(1):716–733. doi:10.31480/2330-4871/203.
- Yin Q, Zheng Y, Wang S, Sun YE, Wang JF, Cheng W. “Postural Alignment First, Symptom Tailored” Integrated Pathway as a Multimodal Peripheral Intervention Strategy for Pain and Cervical Disease (Part 2). Transl Perioper Pain Med. 2025;12(1):734–745. doi:10.31480/2330-4871/204.
- Edinger JD, Arnedt JT, Bertisch SM, et al. Behavioral and psychological treatments for chronic insomnia disorder in adults: an American Academy of Sleep Medicine clinical practice guideline. J Clin Sleep Med. 2021;17(2):255–262. doi:10.5664/jcsm.8988.
- Qaseem A, Kansagara D, Forciea MA, Cooke M, Denberg TD; Clinical Guidelines Committee of the American College of Physicians. Management of chronic insomnia disorder in adults: a clinical practice guideline. Ann Intern Med. 2016;165(2):125–133. doi:10.7326/M15-2175.
- Sateia MJ, Buysse DJ, Krystal AD, Neubauer DN, Heald JL. Clinical practice guideline for the pharmacologic treatment of chronic insomnia in adults: an American Academy of Sleep Medicine clinical practice guideline. J Clin Sleep Med. 2017;13(2):307–349. doi:10.5664/jcsm.6470.
- Riemann D, Baglioni C, Bassetti C, et al. European guideline for the diagnosis and treatment of insomnia. J Sleep Res. 2017;26(6):675–700. doi:10.1111/jsr.12594.
- Liu H, Wu X, Fang X, et al. Effect of perioperative dexmedetomidine on postoperative sleep quality in adult surgical patients: a systematic review and meta-analysis. BMC Anesthesiol. 2023;23:88. doi:10.1186/s12871-023-02048-6.
- Meng D, Mao Y, Song Q, et al. Efficacy and safety of transcutaneous electrical acupoint stimulation (TEAS) for postoperative pain in laparoscopy: a systematic review and meta-analysis of randomized controlled trials. Evid Based Complement Alternat Med. 2022;2022:9922879. doi:10.1155/2022/9922879.
- Morin CM, Vallières A, Guay B, Ivers H, Savard J, Mérette C, Bastien C, Baillargeon L. Cognitive behavioral therapy, singly and combined with medication, for persistent insomnia: a randomized controlled trial. JAMA. 2009;301(19):2005–2015. doi:10.1001/jama.2009.682.
- Brzezinski A, Vangel MG, Wurtman RJ, et al. Effects of exogenous melatonin on sleep: a meta-analysis. Sleep Med Rev. 2005;9(1):41–50. doi:10.1016/j.smrv.2004.06.004.
- Prather AA, Krystal AD, Emsley R, et al. The Effectiveness of Digital Cognitive Behavioral Therapy to Treat Insomnia Disorder in US Adults: Nationwide Decentralized Randomized Controlled Trial. JMIR Ment Health. 2025;12:e84323. doi:10.2196/84323.
- Baethge C, Goldbeck-Wood S, Mertens S. SANRA—a scale for the quality assessment of narrative review articles. Res Integr Peer Rev. 2019;4:5. doi:10.1186/s41073-019-0064-8.
- Page MJ, McKenzie JE, Bossuyt PM, et al. The PRISMA 2020 statement: an updated guideline for reporting systematic reviews. BMJ. 2021;372:n71. doi:10.1136/bmj.n71.
- Buysse DJ, Reynolds CF III, Monk TH, Berman SR, Kupfer DJ. The Pittsburgh Sleep Quality Index: a new instrument for psychiatric practice and research. Psychiatry Res. 1989;28(2):193–213. doi:10.1016/0165-1781(89)90047-4.
- Bastien CH, Vallières A, Morin CM. Validation of the Insomnia Severity Index as an outcome measure for insomnia research. Sleep Med. 2001;2(4):297–307. doi:10.1016/S1389-9457(00)00065-4.
- Johns MW. A new method for measuring daytime sleepiness: the Epworth sleepiness scale. Sleep. 1991;14(6):540–545. doi:10.1093/sleep/14.6.540.
- Nagappa M, Liao P, Wong J, Auckley D, Ramachandran SK, Memtsoudis S, Mokhlesi B, Chung F. Validation of the STOP-Bang questionnaire as a screening tool for obstructive sleep apnea among different populations: a systematic review and meta-analysis. PLoS One. 2015;10(12):e0143697. doi:10.1371/journal.pone.0143697.
- Sweetman A, Lack L, Bastien C. Co-Morbid Insomnia and Sleep Apnea (COMISA): prevalence, consequences, methodological considerations, and recent randomized controlled trials. Brain Sci. 2019;9(12):371. doi:10.3390/brainsci9120371.
- Okajima I, Komada Y, Inoue Y. A meta-analysis on the treatment effectiveness of cognitive behavioral therapy for primary insomnia. J Psychosom Res. 2011;71(2):89–98. doi:10.1016/j.jpsychores.2011.06.010.
- Tan SY, et al. Effects of transcutaneous electrical acupoint stimulation on early postoperative pain and recovery: a systematic review and meta-analysis of randomized controlled trials. Front Med (Lausanne). 2024. doi:10.3389/fmed.2024.1302057.
- Zhang M, et al. Effect of transcutaneous electrical acupoint stimulation on the quality of recovery after surgery: a systematic review and meta-analysis. BMC Anesthesiol. 2024. doi:10.1186/s12871-024-02483-z.
- Xing C, Zhou Y, Xu H, Ding M, Zhang Y, Zhang M, Hu M, Huang X, Song L. Sleep disturbance induces depressive behaviors and neuroinflammation by altering the circadian oscillations of clock genes in rats. Neurosci Res. 2021;171:124–132. doi:10.1016/j.neures.2021.03.006.
- Tripathi R, Das S, Shah S, et al. Opioids and sleep: a review of literature. Sleep Med. 2020;67:269–275. doi:10.1016/j.sleep.2019.06.012.
- Wu Y, Miao Y, Chen X, Wan X. Dexmedetomidine improves postoperative sleep quality in patients undergoing functional endoscopic sinus surgery: a randomized, double-blind, placebo-controlled trial. BMC Anesthesiol. 2022;22:172. doi:10.1186/s12871-022-01711-8.
- Wu J, Jiang S, Xu C, et al. Intranasal dexmedetomidine improves postoperative sleep quality in older patients with chronic insomnia undergoing elective noncardiac surgery: a randomized double-blind placebo-controlled trial. Front Pharmacol. 2023;14:1223746. doi:10.3389/fphar.2023.1223746.
- Alexopoulou C, Kondili E, Diamantaki E, et al. Effects of dexmedetomidine on sleep quality in critically ill patients: a pilot study. Anesthesiology. 2014;121(4):801–807. doi:10.1097/ALN.0000000000000361.
- Liu L, Ma X, Song Z, et al. Effects of melatonin on the prevention of delirium in hospitalized older patients: systematic review and meta-analysis. BMC Pharmacol Toxicol. 2024;25:90. doi:10.1186/s40360-024-00816-9.
- Smith MT, McCrae CS, Cheung J, Martin JL, Harrod CG, Heald JL, Carden KA. Use of actigraphy for the evaluation of sleep disorders and circadian rhythm sleep-wake disorders: an American Academy of Sleep Medicine systematic review, meta-analysis, and GRADE assessment. J Clin Sleep Med. 2018;14(7):1209–1230. doi:10.5664/jcsm.7228.
- Reed JE, Card AJ. The problem with Plan-Do-Study-Act cycles. BMJ Qual Saf. 2016;25(3):147–152. doi:10.1136/bmjqs-2015-005076.
- van de Baan FC, Lambregts S, Bergman E, Most J, Westra D. Involving Health Professionals in the Development of Quality and Safety Dashboards: Qualitative Study. J Med Internet Res. 2023;25:e42649. doi:10.2196/42649.
Table of Contents
- Abstract
- Keywords
- Introduction
- Time Windows and Tools: An Assessment Framework for Perioperative PSD
- Intraoperative phase: Sedation-analgesia structure and CST-DS axis protection
- Phenotype-Guided Nonpharmacologic Interventions: From CBT-I to a CST-DS-Informed Postural Pathway
- Pharmacologic and Anesthetic Strategies: “Minimum Effective Dosing” Through a Mechanistic-Axis Lens
- Digital Phenotyping and Quality Improvement: From Individual Cases to Dashboards
- Discussion
- Conclusion
- Author Contributions
- Conflicts of Interest
- Funding
- AI Use Statement
- Figure 1
- Figure 2
- Figure 3
- Figure 4
- Table
- Table
- References