Translational Perioperative and Pain Medicine (ISSN: 2330-4871)
ARTICLE DOI: 10.31480/2330-4871/212
Research Article | Volume 12 | Issue 1 Open Access
Perioperative Sleep Disturbances: A Multi-Axis, Hypothesis-Driven Classification Framework (Part I: Integrating the CSDS/CST-DS Axis with a Five-Axis Mechanistic Model)
Qin Yin1#, Fei-Fei Lu2#, Ming-Yue Cheng1, Yu-E Sun3, Jin-Feng Wang4* and Wei Cheng5*
1The Affiliated Hospital of Xuzhou Medical University, Xuzhou 221002, P.R. China
2The TCM Hospital Affiliated to Nanjing University of Chinese Medicine, Xuzhou 221009
3The Affiliated Hospital of Nantong University, No. 20 Xisi Road, Nantong, China
4The Suzhou Hospital of the Chinese Academy of Traditional Chinese Medicine - Xiyuan Hospital, Suzhou, China
5The Affiliated Huai'an No.1 People's Hospital of Nanjing Medical University, Huai'an 223300, P.R. China
#Contributed equally to this work
Wei Cheng, Huai'an No.1 People's Hospital of Nanjing Medical University, Huai'an 223300, China, Tel: +8618796205791, E-mail: 53974314@qq.com; Jin-Feng Wang, The Suzhou Hospital of the Chinese Academy of Traditional Chinese Medicine - Xiyuan Hospital, P.R. Suzhou, China, Tel: +8618168779112, E-mail: 709034013@qq.comEditor: Renyu Liu, MD; PhD; Professor, Department of Anesthesiology and Critical Care, Perelman School of Medicine at the University of Pennsylvania, Center of Penn Global Health Scholar, 336 John Morgan building, 3620 Hamilton Walk, Philadelphia, PA 19104, USA, Fax: 2153495078, E-mail: RenYu.Liu@pennmedicine.upenn.edu
Received: Nov 17, 2025 | Accepted: Jan 14, 2026 | Published: Jan 17, 2026
Citation: Yin Q, Lu FF, Cheng MY, et al. Perioperative Sleep Disturbances: A Multi-Axis, Hypothesis-Driven Classification Framework (Part I: Integrating the CSDS/CST-DS Axis with a Five-Axis Mechanistic Model). Transl Perioper Pain Med 2026; 12(1):807-818
Abstract
Background: Perioperative sleep disturbances (PSD) are common and are associated with a range of adverse postoperative outcomes. However, the prevailing “riskdomain–symptom-spectrum” paradigm mainly provides a global risk description and often lacks a mechanistic, bedside-operational structure for identifiable trigger chains.
Objective: To propose and justify incorporating cervicogenic sleep disorder syndrome (CSDS) and its perioperative operational mechanistic axis-cervical spine-triggered sleep disturbance (CST-DS)-into PSD classification, and to generate a multi-axis framework with falsifiable research predictions.
Methods: Using a narrative integrative review approach, we performed thematic searches and citation chaining across interdisciplinary PSD-related evidence. We propose a fiveaxis mechanistic model and present a tiered qualitative appraisal of evidence strength for each axis. Based on this synthesis, we derive candidate composite phenotypes and specify boundary conditions and clinical practice mapping.
Results: We propose that five mechanistic axes jointly define perioperative sleep phenotypes. Axis 1 is the newly introduced CST-DS postural-ventilatory trigger axis. Axes 2-5 cover (respectively) inflammation-immune/ neuroinflammation and brain clearance–related pathways, autonomic-endocrine stress, pharmacologic-neural network mechanisms, and environment–process–circadian factors. We further derive six candidate composite phenotypes (Type 1-6) intended for stratified enrolment and interactioneffect testing, and we outline key differential points to reduce misattribution.
Conclusions: This framework is a hypothesis-generating, multi-axis classification model. Its core value lies in translating a bedside-recognizable “cervical-postureventilation-autonomic” trigger chain into an operational mechanistic axis, thereby enabling preregisterable and falsifiable clinical predictions and study-design elements. Part II will translate the framework into bedside screening checklists and stratified intervention pathways and will propose a validation agenda.
Keywords
Perioperative sleep disturbances, Cervicogenic sleep disorder syndrome, Postoperative complications, Multi-axis model, CSDS/CST-DS
Key Terms and Abbreviations: PSD: Perioperative Sleep Disturbances-refers to insomnia-like symptoms, sleep architecture/circadian disruption, and sleep-disordered breathing (SDB) occurring in the perioperative period. Note: In this manuscript, PSD = perioperative sleep disturbances; it does not refer to “psychosocial disorder”; CSDS: Cervicogenic Sleep Disorder Syndrome-a hypothesis-driven framework emphasizing that cervical structural/functional abnormalities and cervicoscapular soft-tissue dysfunction may constitute important triggers or vulnerability factors for sleep disturbance; CST-DS axis: Cervico-ScapuloThoracic-Diaphragmatic-Sympathetic (CST-DS)-a trigger dimension describing the “cervical-scapular-thoracic cagediaphragm-sympathetic” chain, highlighting how cervical load and postural abnormalities may influence sleep via ventilatory mechanics, autonomic regulation, and coupled reflex pathways; COMISA: Co-Morbid Insomnia and Sleep Apnea; Mechanistic axis: A mechanistic dimension within the multi-axis model- combinations across axes are used to characterize PSD heterogeneity and generate testable mechanistic hypotheses; Phenotypes (Type 1-6): Candidate composite phenotypes formed by different combinations of dominant and secondary axes across the five mechanistic axes; used for risk stratification, differential inference, and stratified study design. Note: Type 1-6 are the unified naming system used in this manuscript. They are intended to be testable/falsifiable and should not be interpreted as established diagnostic conclusions.
Introduction
Perioperative sleep disturbances (PSD) are highly prevalent in surgical populations and are associated with adverse outcomes such as postoperative pain, anxiety, delirium, infection, and reduced quality of recovery [1-5]. Prior narrative and systematic reviews commonly adopt a “risk-domain-symptom-spectrum” framework. Risk domains include psychological/emotional factors, surgery and pain, anesthesia/analgesic medications, inflammatory-immune responses, and environmental and care-process factors; symptom spectra encompass insomnia, sleep fragmentation, circadian disruption, and sleep-disordered breathing (SDB) [1-4]. This framework is useful for screening and risk stratification, but it provides limited structured integration of bedside-identifiable triggers that may reflect etiologic chains-for example, coupling among the cervical spine, posture, ventilation, and autonomic arousal [6-8].
To complement the “risk-domain-symptom-spectrum” approach with a more bedside-actionable mechanistic chain, we propose two closely linked concepts: Cervicogenic sleep disorder syndrome (CSDS) and the cervical spine-triggered sleep disturbance (CST-DS) axis. CSDS is a hypothesis-based etiologic framework suggesting that cervical spine structural-functional abnormalities and related neuromuscular-fascial chain imbalance may constitute an “at-risk substrate/trigger context” for sleep disturbance in selected populations [9,10]. CST-DS is the perioperative operationalization of this construct as a mechanistic axis: it emphasizes that cervical load and postural compensation can alter cervico-thoracic-diaphragmatic coordination and upper-airway mechanics/respiratory control stability, and can couple with a high-arousal autonomic state, thereby increasing sleep fragmentation, arousals, or vulnerability to SDB. A key feature is that CST-DS can be identified via bedside cues (neck/shoulder discomfort, postural abnormalities, posture-related dyspnea, and exposures that increase cervical load during specific positions or intubation), and it can be translated into falsifiable predictions in research [9-11].
A schematic of the anatomic-functional coupling chain underlying CSDS and the CST-DS axis is shown in figure 1. This coupling is presented to generate testable hypotheses and should not be interpreted as an established causal chain.
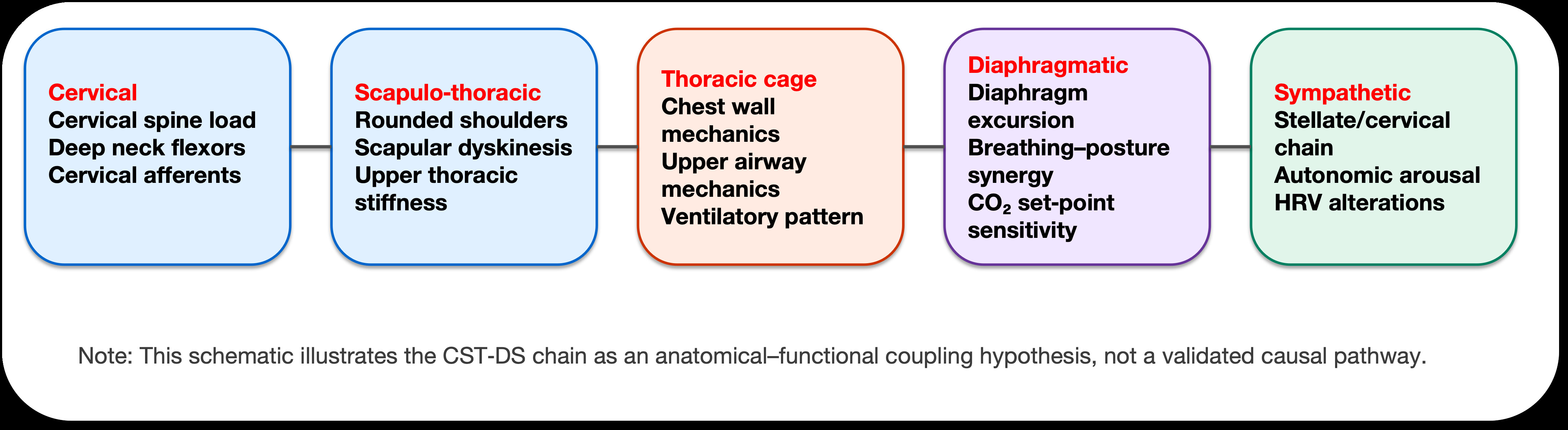
Figure 1: CST‑DS anatomical-functional coupling schematic (conceptual).
This schematic summarizes the CST-DS chain as an anatomical-functional coupling hypothesis, linking cervical load and neck-shoulder mechanics to ventilatory patterning and autonomic arousal. It is intended for hypothesis generation, not as a validated causal pathway or clinical decision algorithm.
Abbreviations: CO2: Carbon dioxide; CSDS: Cervicogenic Sleep Disorder Syndrome; CST-DS: Cervico-Scapulo-Thoracic-Diaphragmatic-Sympathetic (CST-DS) axis; HRV: Heart Rate Variability<
This article addresses the following question: Can a cervical spine–posture–related trigger mechanism be embedded as an operational “etiologic axis” in PSD classification to improve perioperative risk stratification, differential diagnosis, and stratified enrollment for research? Because CSDS/CST-DS remains an emerging, hypothesis-generating construct, we use a narrative integrative review to align cross-disciplinary evidence and present evidence strength in a tiered manner in the text and in Table 1, aiming to reduce the risk of over-causal language [6-8].
To address gaps in existing frameworks, we previously proposed CSDS as a hypothesis-based etiologic construct and delineated A-J subtypes to reframe explanatory and interventional pathways for selected refractory insomnia and SDB [9,10]. Cervicogenic headache, as a secondary headache driven by cervical structural–functional abnormalities, has been summarized in systematic reviews and provides an analogy supporting cautious transfer of the “cervicogenic” concept to sleep disorders [11]. This series is presented in two parts: Part I introduces the integration of the CSDS/CST-DS axis into a multi-axis mechanistic model; Part II will provide bedside assessment and stratified intervention pathways and will discuss validation designs suitable for preregistration.
Scope, relationship to prior work, and methodological notes
This is a conceptual, hypothesis-driven review intended to propose a testable multi-axis classification framework rather than to deliver “proven conclusions” in the manner of a formal systematic review [6-8]. Relative to prior discussions of the CSDS construct, selected subtypes, and the “Postural Alignment First, Symptom Tailored” pathway [9,10], the incremental contributions of the present manuscript are: (1) explicitly defining CSDS/CST-DS as an operational mechanistic axis and integrating it into the main PSD mechanistic domains within a shared coordinate system; (2) proposing a five-axis model with qualitative evidence-strength grading (Table 1) [9-36]; and (3) specifying boundary conditions and differential points to reduce the risk of over-attributing sleep disturbance to cervicogenic mechanisms. Our search strategy combined thematic searching with citation chaining, focusing on PSD/sleep architecture changes, cervical spine-posture-ventilatory mechanics, neuroinflammation/delirium, and glymphatic-like clearance. We searched PubMed, Embase, and Web of Science for English human studies from 2015-2025 and supplemented key foundational papers for conceptual provenance; guidelines/systematic reviews, RCTs, and larger cohorts were prioritized, while animal/in-vitro studies were used only as explanatory background [6-8]. In alignment with TPPM transparency expectations, the submission and cover letter disclose potential overlap with prior work and state that figures 1-5 were created or redrawn as original materials [9,10].
Table 1: Qualitative grading of evidence strength for the five mechanistic axes.
|
Mechanistic axis |
Core mechanism (overview) |
Common perioperative evidence types |
Evidence strength (this manuscript) |
|
Axis 1: Posture-ventilation (CSDS/CST-DS) |
Cervical/postural dysfunction, altered upper-airway mechanics, ventilatory instability, and cervicosympathetic and vagal reflex coupling (CSDS/CST-DS as a “trigger axis/etiologic subset”) [9-20]. |
Predominantly observational studies and mechanistic extrapolation; limited perioperative randomized trials directly targeting “cervicogenic drivers” [9-20]. |
Emerging |
|
Axis 2: Inflammation-immune-CSF/venous outflow-glymphatic-like clearance |
Systemic inflammation, endothelial dysfunction, altered BBB permeability, CSF/glymphatic-like clearance, and links between neuroinflammation and PND/delirium risk [21-34]. |
Population-level evidence linking inflammation-sleep-delirium/PND is comparatively strong; glymphatic-like evidence is expanding but remains largely indirect/extrapolated in the PSD context [21-34]. |
Moderate (glymphatic-like: emerging) |
|
Axis 3: Autonomic-endocrine stress |
Sympathetic-vagal imbalance, activation of stress axes (e.g., HPA), and circadian regulatory dysregulation (a hyperarousal/vulnerability background) [1-5,35,36]. |
Commonly studied in perioperative stress responses and sleep-architecture changes; some interventional evidence suggests modifiability, but heterogeneity is substantial [1-5,35,36]. |
Moderate |
|
Axis 4: Pharmacologic-neural network |
Effects of anesthetic/analgesic/sedative agents on sleep architecture, arousal networks, and respiratory drive (including contributions to SDB risk and sleep fragmentation). [1-2,4] |
Substantial evidence linking perioperative medications to sleep architecture, delirium, and respiratory events; mechanistic granularity and inter-individual variability remain to be refined [1-2,4]. |
Established |
|
Axis 5: Environment-process-circadian |
Light exposure, noise, nursing workflow, activity-social rhythms, and the inpatient environment leading to circadian disruption and sleep fragmentation [1-4]. |
Many studies on environmental and circadian-optimization interventions, but implementation heterogeneity is large and effects are population- and process-dependent [1-4]. |
Moderate |
Existing approaches to classifying perioperative sleep disturbances: From risk domains to symptom spectra
Most studies treat perioperative sleep disturbances (PSD) as a multifactorial syndrome. Commonly cited risk domains include psychological and emotional factors, pain and surgical stress, anesthetic/analgesic medications, inflammatory-immune responses, pre-existing sleep disorders/comorbidities, and environmental and care-process factors [1-5]. Perioperative cohort studies further suggest that preoperative sleep fragmentation or increased nocturnal awakenings are associated with a higher risk of postoperative delirium, implying that sleep may be a modifiable vulnerability factor for perioperative neurocognitive disorders (PND) [37,38]. Although the “risk-domain-symptom-spectrum” framework is practical for screening and broad risk stratification, it remains largely correlational and offers limited structure for bedside-identifiable, triggerable etiologic chains (e.g., cervical spine-posture-ventilation-autonomic coupling). For this reason, we propose a “five-axis mechanistic model” and subsequently derive candidate phenotypes by combining axes, providing an entry point for mechanistic hypotheses and stratified study design.
An overview of the PSD timeline, major clinical outcomes, and traditional risk domains is provided in figure 2. The purpose of this figure is to establish clinical context so that the subsequent “mechanistic axes-phenotypes” framework can be anchored to time-window-based management.
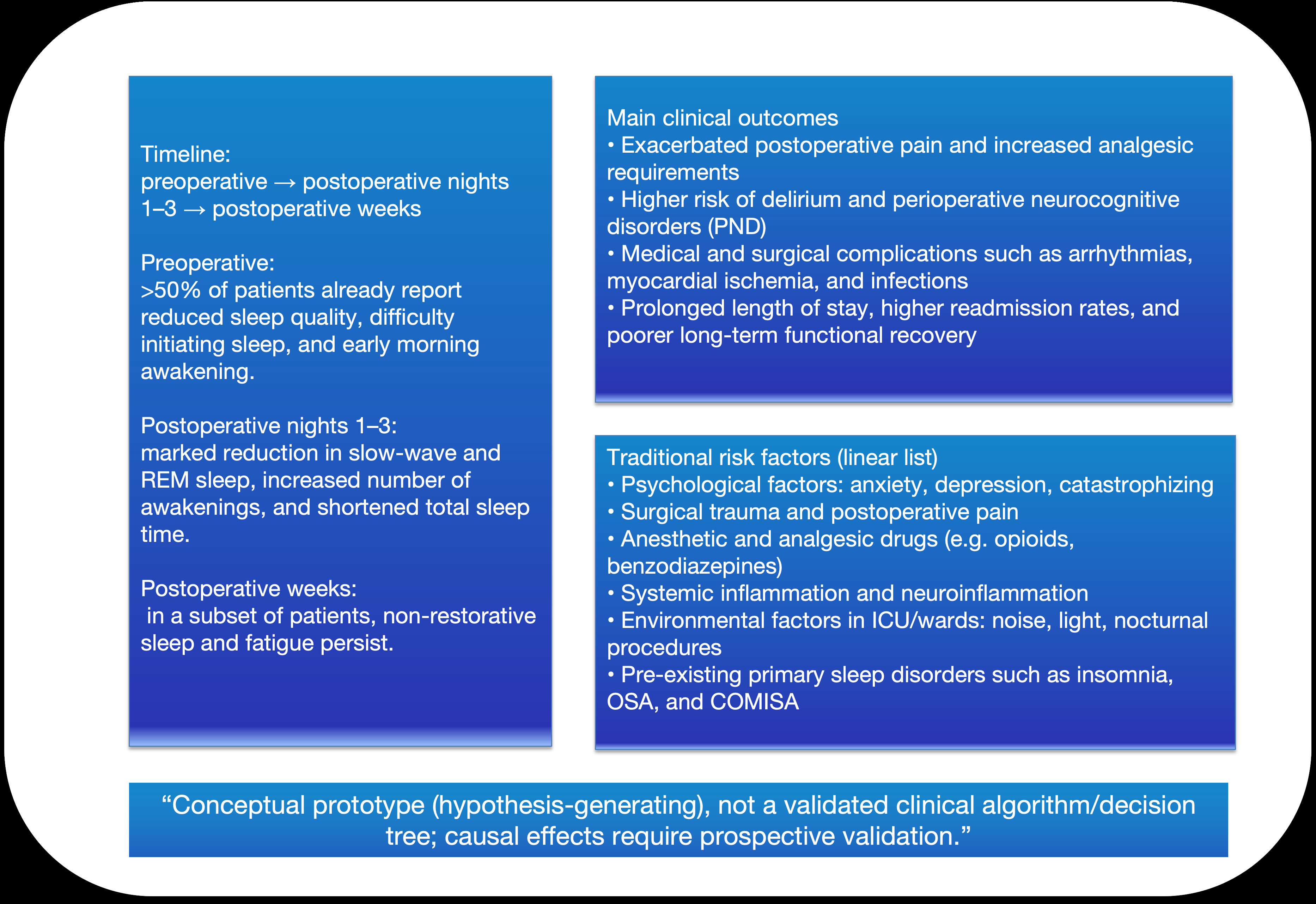
Figure 2: Perioperative timeline and clinical consequence space of perioperative sleep disturbance (PSD) (conceptual prototype).
This panel summarizes the perioperative timeline and clinical consequence space of PSD, including typical sleep-architecture changes across perioperative phases and downstream outcomes (pain, PND, medical/surgical complications, utilization) alongside traditional risk factors. It is a conceptual prototype used to frame research questions and stratification logic.
Abbreviations: COMISA: Comorbid Insomnia and Sleep Apnea; ICU: Intensive Care Unit; OSA: Obstructive Sleep Apnea; REM: Rapid Eye Movement Sleep; PND: Perioperative Neurocognitive Disorders; PSD: Perioperative Sleep Disturbance
CSDS and the CST-DS axis: A cervical-to-sleep pathophysiologic cascade
CSDS considers cervical spine structural-functional abnormalities and the resulting neuromuscular-fascial chain imbalance as potential triggers for certain sleep disturbances. To avoid over-causal phrasing, we summarize the proposed cascade as “possible associations/possible contributions”:
- Postural compensation (forward head posture, rounded shoulders, and thoracic kyphosis) may be associated with poorer subjective sleep quality and may affect respiratory mechanics through impaired cervico-thoracic-diaphragmatic coordination [12].
- Cervical structural/functional abnormalities may be associated with neck-shoulder pain or discomfort and may further promote compensatory postural patterns [13,14].
- Upper-airway mechanics and instability of respiratory control may be associated with sleep fragmentation, increased arousals, or a higher risk of sleep-disordered breathing (SDB).
Additional supporting evidence: Beyond cross-sectional associations, some interventional studies suggest that treatments targeting posture and/or cervical function may improve sleep and quality of life, providing early external support for the “intervenability” of the CST-DS axis [15]. In addition, sleep position can influence neck muscle activity, offering a biomechanical rationale for a “sleep posture–cervical load–sleep continuity” linkage [16].
Clinical studies have also reported effects of cervical support and pillow height on cervical alignment and subjective sleep, which can serve as analog evidence for perioperative “posture-cervical load management” [17,18]. Sympathetic-vagal imbalance may couple with inflammatory responses and manifest as heightened nocturnal arousal and poor recovery. Perioperative exposures (e.g., intubation, prone or hyperextended positioning, cervical immobilization, traction) may transiently amplify these chains, thereby increasing vulnerability to the onset and persistence of PSD [19,20,35].
Boundary conditions and key points for differential diagnosis
Within this framework, CSDS is positioned as an etiologic subset/trigger axis for PSD-intended to explain “why this patient is more vulnerable to a particular sleep problem in the perioperative setting” and “which trigger chains might be prioritized for intervention,” rather than to replace standard sleep-medicine diagnoses such as obstructive sleep apnea (OSA) or primary insomnia [1-4]. Accordingly, when dominant factors are present-clear high OSA risk, medication-related sleep-architecture changes, severe pain, psychiatric/psychological conditions, or systemic disease (e.g., heart failure, thyroid dysfunction)-evaluation and management should first follow conventional pathways. CST-DS-trigger clues (cervical spine-posture-ventilation-autonomic coupling) can then be assessed to refine risk stratification and personalize interventions [1-5].
Bidirectional sleep–pain interactions and neuroimmune vulnerability pathways have been repeatedly documented, suggesting that some patients may exhibit coupling between “sleep vulnerability” and risk of persistent pain [5,35,36]. In practice, we recommend first completing symptom assessment and stratification using scales such as the PSQI and ISI, then moving to mechanistic-axis clue identification to reduce misattribution. For suspected OSA, tools such as STOP can be used for stratification, and complication risk should be interpreted in light of prior evidence [39-42].
Perioperative intubation, cervical immobilization/traction, and special positioning can impose substantial short-term cervical load. If postoperative symptoms include difficulty initiating sleep, increased awakenings, morning headache/neck-shoulder discomfort, or posture-related breathing discomfort, the CST-DS axis can be incorporated as an explanatory frame and used to generate testable hypotheses [12-18]. Compared with related concepts, the insomnia-pain model emphasizes broadly applicable mutual reinforcement mechanisms [5,35], whereas CSDS/CST-DS emphasizes a specific “cervical load-posture-ventilation-autonomic” trigger axis that is more likely to contribute to PSD vulnerability when posture/cervical-load sensitivity features are present [12-18]. Central sensitization is better considered a background vulnerability dimension within Axes 2/3 rather than a prerequisite for Axis 1 to be relevant [35,36]. Overall, CSDS should be treated as a complementary trigger axis that enhances differential diagnosis and stratified intervention precision on top of standard diagnostic systems [1-4,41,42].
Transparency statement on potential overlap and figure/table reuse
This manuscript necessarily continues certain conceptual background elements from our prior work (e.g., the definition/classification of CSDS and the “Postural Alignment First” pathway). However, the incremental contributions of the present article focus on: placing CSDS/CST-DS as one of five mechanistic axes for perioperative PSD within a unified coordinate system, and using this structure to propose a workflow-based derivation of candidate types (Type 1-6), evidence-strength grading, and explicit boundary conditions. We will clearly describe the relationship to prior work and any potential areas of overlap in the submission system and in the cover letter to ensure transparency and traceability. Figures 1,2,3,4 and 5 and tables 1,2 and 3 are intended to be newly created or redrawn as original materials; if reuse of any identifiable element is required (including structure, layout, or components), we will obtain the necessary permissions in accordance with journal and publisher policies and provide appropriate source and permission statements in the figure legends.
From Five Mechanistic Axes to Actionable Phenotypes: Evidence Gradients, Workflow-Based Derivation, and Candidate Types (Type 1-6)
How evidence is translated into phenotypes: The role and reading order of three tables
Prior reviews consistently indicate that PSD is driven by multiple factors and is highly heterogeneous. Although the traditional “risk-domain-symptom-spectrum” approach is useful for screening and stratification, it offers limited support for mechanism-oriented phenotyping and stratified enrollment in research [1-4], and the bidirectional relationship between sleep and pain represents an important, broadly relevant background dimension 5]. Accordingly, this narrative integrative review treats combinations of axes as the defining elements of phenotypes, and Table 1 presents a qualitative, tiered grading of evidence strength for each axis [6-8].
At the evidence level, perioperative human studies and reviews have increasingly documented links between sleep disturbance and PND/delirium, alongside accumulating evidence related to neuroinflammation and blood–brain barrier (BBB) changes [21-25]. Interventional studies centered on sleep promotion and circadian optimization also suggest potential effects on delirium and related outcomes [26,27]. In parallel, foundational work and subsequent reviews on sleep and cerebrospinal fluid (CSF) exchange/glymphatic-like clearance provide a traceable biological backdrop for an “inflammation-immune-BBB-CSF/venous-glymphatic-like” mechanistic domain [28-32]. Animal data under anesthesia further suggest possible coupling with electrophysiologic and autonomic markers [33], and integrative perioperative perspectives have proposed that neuroinflammation and glymphatic-like dysfunction may jointly contribute to PND [34]. From a clinical standpoint, we recommend completing baseline stratification at the “symptom level” and “risk level” using instruments such as PSQI/ISI and OSA screening tools (e.g., STOP) before moving to mechanistic-axis clue identification, to reduce misattribution [39-42]. Within this framework, Axis 1 (the postural-ventilatory axis) is the new, operational etiologic axis introduced in this series, conceptually grounded in CSDS and the “Postural Alignment First” pathway and informed by the analog framework of cervicogenic disorders [9-11].
- Axis 1: CSDS/CST-DS postural–ventilatory axis [9-11,12-18].
- Axis 2: inflammation–immune–BBB–CSF/glymphatic-like axis [21-34].
- Axis 3: autonomic–endocrine stress axis [1-4,[35,36].
- Axis 4: pharmacologic–neural network axis [2,4].
- Axis 5: environment–process–circadian axis [1-4].
To integrate CSDS/CST-DS with established PSD frameworks, we propose a multi-axis model composed of five mechanistic axes (Table 1). An overview of the five axes and their relationship to the CST-DS axis is provided in figure 3. Subsequent phenotype derivation uses this axis space to determine primary versus secondary axes and to define axis combinations.
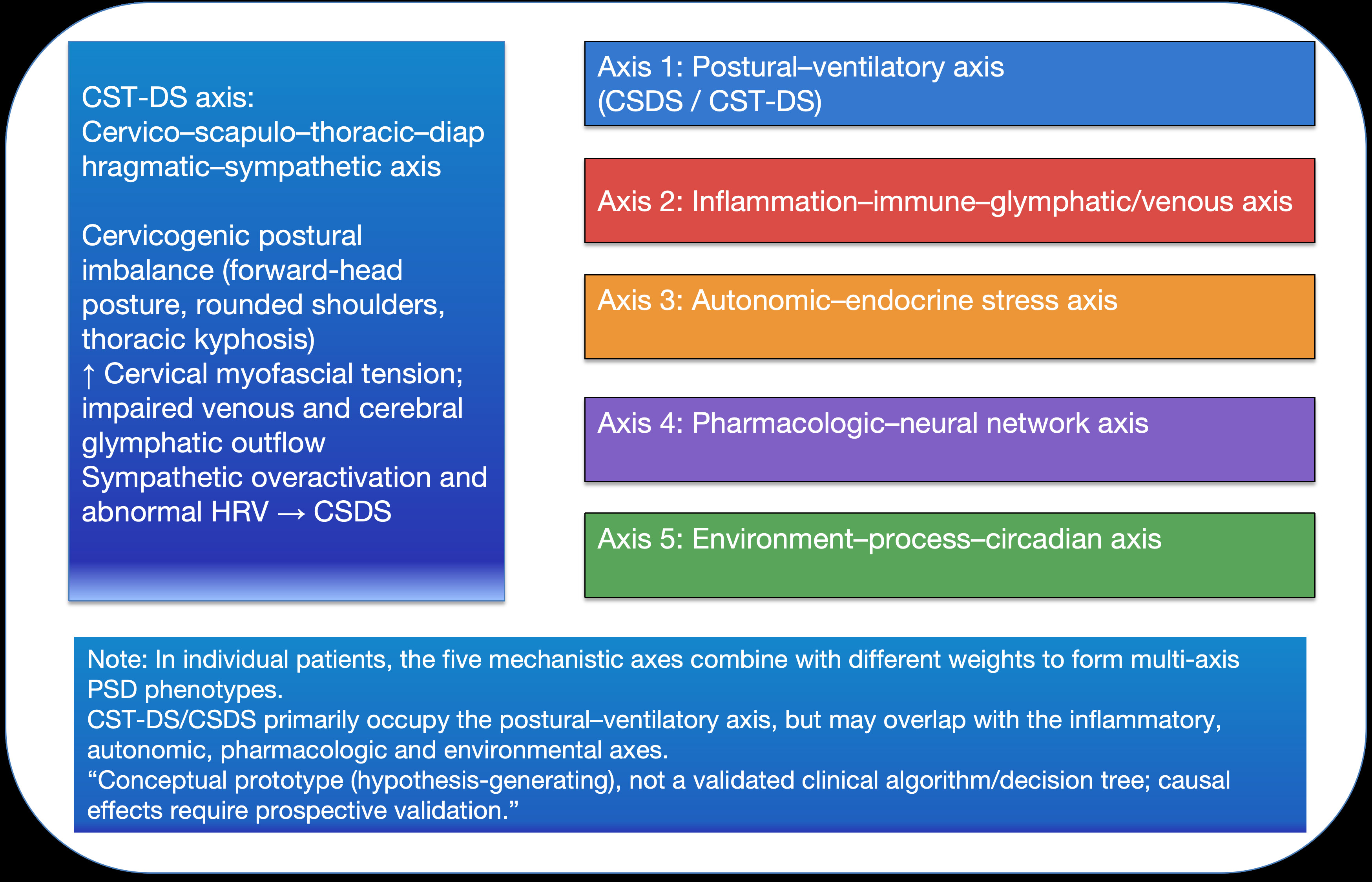
Figure 3: CST-DS/CSDS mapped onto a five-axis mechanistic framework for multi-axis PSD phenotype construction (conceptual).
This figure maps CST-DS/CSDS into a five-axis mechanistic space and illustrates how different axes can combine with varying weights to yield multi-axis PSD phenotypes. The purpose is to operationalize mechanistic thinking for phenotype derivation and downstream pathway alignment.
Abbreviations: CSDS: Cervicogenic Sleep Disorder Syndrome; CST-DS: Cervico-Scapulo-Thoracic-Diaphragmatic-Sympathetic (CST-DS) axis; HRV: Heart Rate Variability; PSD: Perioperative Sleep Disturbance
Note: The strength of supporting evidence is not uniform across axes. Table 1 is intended to transparently display the evidence gradient and to avoid presenting hypothesis-level mechanisms as settled conclusions. In the perioperative PSD context, CST-DS-related mechanisms and glymphatic-like/CSF-related pathways are still largely supported by indirect or extrapolated evidence and are therefore categorized as “emerging evidence” [9-11,12-20,28-34].
The purpose of Table 1 is to “put the evidence gradient on the table first.” Because direct perioperative evidence is uneven across the five axes, no classification should treat emerging mechanisms as definitive. Instead, uncertainty should be made explicit through evidence-strength labeling. With that premise, the next step is not simply to list more risk factors, but to translate “risk-domain information” into repeatable bedside decision steps, thereby reducing interpretive divergence across disciplines for the same patient. To that end, we operationalize phenotype identification into a four-step workflow, P1-P4 (Table 2): First complete baseline stratification at the symptom level and OSA-risk level, then identify CST-DS trigger clues, next assign primary versus secondary axes among the five, and finally generate a phenotype label suitable for research stratification.
Table 2: Workflow-based phenotype derivation (P1-P4), mapped to Type 1-6.
|
Step (P1-P4) |
Purpose |
Key inputs/decisions |
Outputs (mapping to Type 1-6) |
References |
|
P1: Baseline screening |
Define the PSD “entry problem” and capture conventional risk domains. |
Pre-/postoperative sleep complaints; PSQI/ISI stratification [39,40]. OSA risk screening (STOP questionnaire) [41]. Conventional risk-domain data including pain, anxiety/hyperarousal, medications, and environmental exposures [1-5]. Incorporate existing evidence on OSA-related perioperative complication risk [42]. |
Clarify the dominant presentation (insomnia-like, circadian disruption, SDB, etc.) and select the priority axis/axes for P2-P3. |
|
|
P2: Identify Axis 1 cues |
Detect CST-DS trigger cues and posture-ventilation coupling signals. |
Postural cues (forward head posture, rounded shoulders, and thoracic kyphosis) associated with subjective sleep quality [12]. Cues linking neck pain/cervicoscapular discomfort to sleep disturbance (including longitudinal evidence) [13,14]. Sleep posture effects on cervical muscle activity/load [16]. Effects of pillows/cervical support on cervical alignment and/or subjective sleep [17-20]. In addition, incorporate perioperative exposures such as tracheal intubation, prolonged or fixed positioning, traction, and cervical hyperextension/hyperflexion, and any temporally related symptom onset as supportive cues within the Axis 1 trigger hypothesis [9,10]. |
If Axis 1 cues are strong → prioritize Type 4 as a candidate; if absent/weak → proceed to P3 with other axes as dominant drivers. |
|
|
P3: Determine dominant vs secondary axes |
Translate risk-domain information into axis weighting and generate testable hypotheses. |
Integrate five-axis evidence and available bedside indicators (e.g., inflammatory markers when available, medication history, environment/process exposures, and autonomic cues such as HRV). Preoperative sleep disruption linked to postoperative delirium/PND risk [12,13]. Evidence chain for perioperative neuroinflammation/BBB changes and delirium/PND [21,27]. Foundational studies, reviews, and anesthesia-related evidence on glymphatic-like/CSF clearance [28-34]. Incorporate sleep-pain-neuroimmune susceptibility pathways as background vulnerability cues [35,36]. |
Assign dominant-axis combinations and map to Type 1-6 (e.g., Axis 3 dominant → Type 1; Axis 4 dominant → Type 2; Axis 2 dominant → Type 3; Axis 1 dominant → Type 4; Axis 5 dominant → Type 5; multi-axis overlap → Type 6/Complex). |
|
|
P4: Output phenotype and stratify enrollment |
Produce an operational phenotype label linked to intervention and study design. |
Map P3 dominant/secondary axes to Type 1-6; record boundary conditions and differential considerations. Maintain priority for standard pathways for OSA/insomnia (PSQI/ISI and STOP screening) [39-42]. Interpret Axis 1 (CST-DS) as a posture- and cervical-load-sensitive trigger/vulnerability axis that complements-not replaces-standard diagnosis [9,10]. |
Output Type 1-5 or multi-axis overlap Type 6 (Complex/COMISA/overlap) for stratified RCTs or prospective cohort enrollment and interaction-effect testing. |
Table 2’s P1-P4 is not presented as a “new diagnostic pathway.” Rather, it is a minimal workflow that converts mechanistic hypotheses into operational inputs and outputs. Its output is a candidate phenotype label to support stratified management and research enrollment, not to replace standard diagnostic systems for OSA, primary insomnia, and related disorders. To help readers apply the workflow quickly in both clinical and research settings, we further compress the P1-P4 outputs into six candidate composite phenotypes (Table 3). Each type is indexed by its dominant axis and is accompanied by a minimal assessment set and falsifiable predictions, serving as a “quick reference / stratification scaffold” aligned with the axis-weight matrix in figure 4 and subsequent intervention modules.
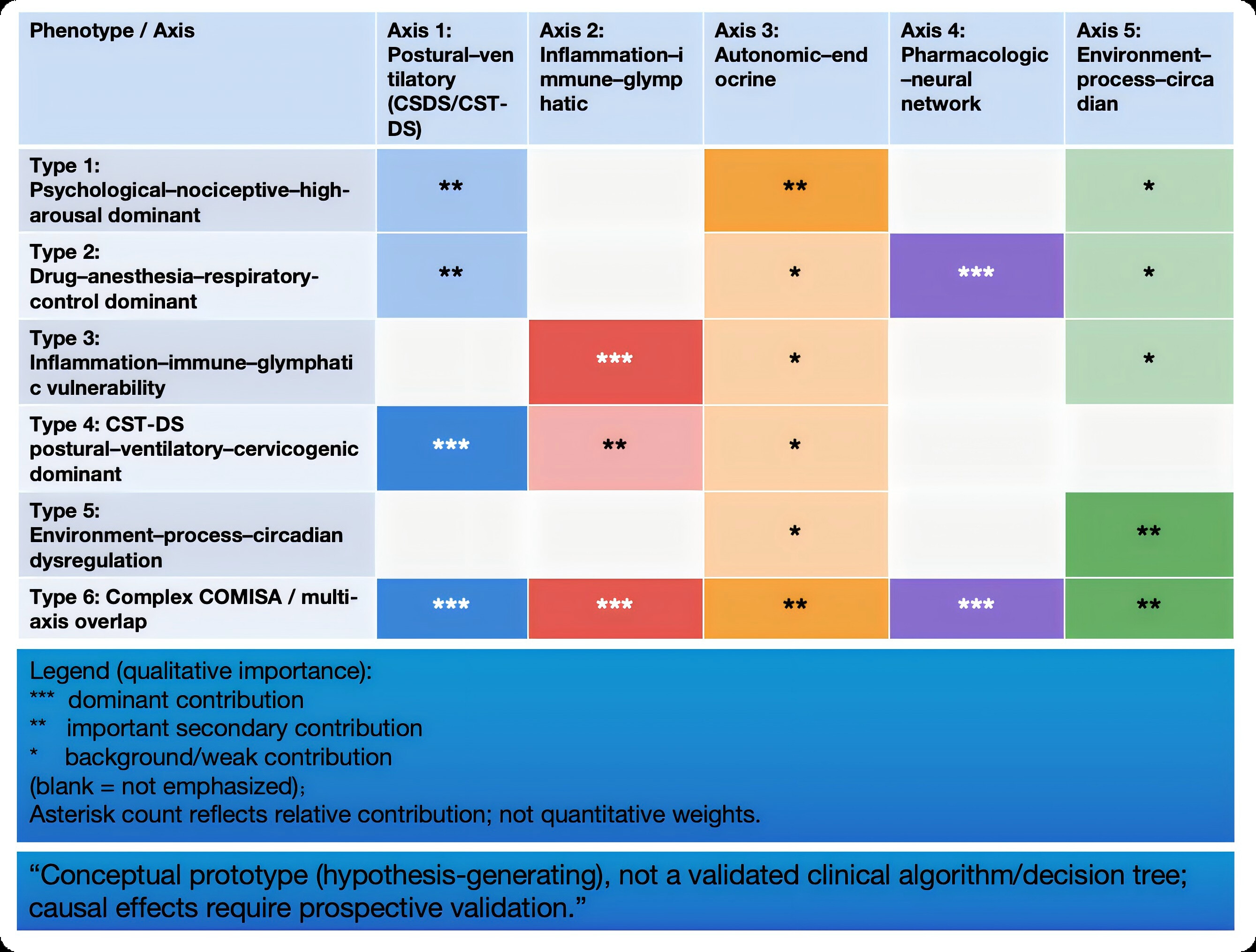
Figure 4: Qualitative mechanistic axis–phenotype importance matrix (asterisk-coded, non-quantitative) (conceptual).
Qualitative axis-phenotype matrix. Asterisk count indicates relative contribution of each mechanistic axis to a given phenotype (*** dominant; ** important secondary; * background/weak; blank = not emphasized). This is a qualitative visualization and does not represent quantitative weights; prospective validation is required.
Abbreviations: COMISA: Comorbid Insomnia and Sleep Apnea; CSDS: Cervicogenic Sleep Disorder Syndrome; CST-DS: Cervico-Scapulo-Thoracic-Diaphragmatic-Sympathetic (CST-DS) axis; OSA: Obstructive Sleep Apnea; PSD: Perioperative Sleep Disturbance
For rapid bedside execution, a schematic of the P1-P4 workflow is provided in figure 5
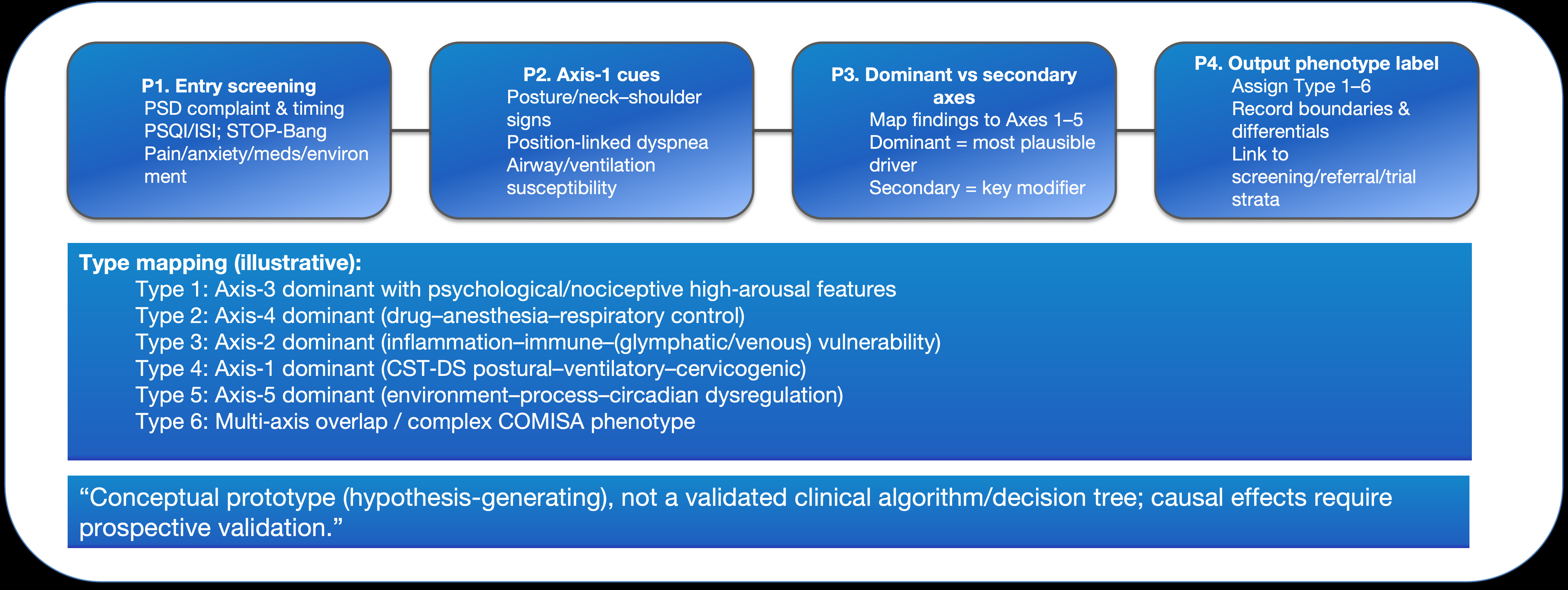
Figure 5: Phenotype derivation workflow (P1-P4).
Phenotype derivation workflow (P1-P4) aligned to Type 1-6. The workflow provides a minimal input-output process to translate mechanistic hypotheses into candidate phenotype labels for stratified management or research enrollment. It is a conceptual prototype, not a validated clinical algorithm/decision tree.
Abbreviations: COMISA: Comorbid Insomnia and Sleep Apnea; CST-DS: Cervico-Scapulo-Thoracic-Diaphragmatic-Sympathetic (CST-DS) axis; ISI: Insomnia Severity Index; QI: Quality Improvement; STOP-Bang: STOP-Bang Questionnaire (Screening Tool for Obstructive Sleep Apnea); P1: Step 1 (Entry screening); P2: Step 2 (Axis-1 cues); P3: Step 3 (Dominant vs secondary axes); P4: Step 4 (Output phenotype label); PSD: Perioperative Sleep Disturbance; PSQI: Pittsburgh Sleep Quality Index
Operational definitions and the minimal assessment package for the six candidate composite phenotypes are provided in Table 3.
A matrix summary of each phenotype’s dominant mechanistic axes (qualitative weights) is presented in Figure 4 to enable one-to-one alignment with the Table 3 definitions.
Table 3: Candidate composite phenotypes (Type 1-6): Definitions, minimum assessment, and falsifiable predictions (aligned with figure 4 and table 2).
|
Type |
Dominant axis |
Core clinical picture (clues) |
Minimum assessment (prioritize P1/P2) |
Priority intervention module / falsifiable prediction |
|
Type 1: Hyperarousal/stress-dominant |
Axis 3 (± Axis 5/4) |
Hyperarousal, difficulty initiating sleep, increased nocturnal awakenings; may include circadian fluctuation [2,4]. |
HRV/resting heart rate suggesting increased sympathetic tone; PSQI/ISI stratification [39,40]. |
Circadian and stress-modulation strategies should improve hyperarousal and HRV metrics more than general measures [2,4]. |
|
Type 2: Medication–anesthesia-respiratory-control dominant |
Axis 4 (± OSA/COMISA) |
Residual effects of opioids/benzodiazepines/sedatives → disrupted sleep architecture and impaired respiratory drive [2,4]. |
Medication review; STOP screening for OSA risk and stratify accordingly [41,42]. |
Optimizing analgesia-sedation regimens yields more pronounced short-term PSD improvement [2,4]. |
|
Type 3: Inflammation/neuroinflammation-dominant |
Axis 2 |
Prominent inflammatory response; increased delirium/PND risk; cues linking preoperative sleep fragmentation to delirium risk [12,13]. |
Infection/inflammation risk signals plus inflammatory markers when feasible; concurrently monitor neurocognitive outcomes [21,23]. |
Anti-inflammatory and recovery-promoting strategies should exert stronger effects on sleep fragmentation and delirium-related outcomes [21,27]. |
|
Type 4: CST-DS/posture-ventilation dominant |
Axis 1 (± Axis 3) |
Neck/shoulder discomfort, postural abnormalities, posture-related breathing discomfort; intubation and special/prolonged positioning exposures may amplify symptoms [9,10]. |
Screen posture and positioning-exposure history; assess sleep posture/support when indicated; run PSQI/ISI in parallel to reduce misattribution [39,40]. |
Targeted posture–ventilation interventions (positioning and cervical-load management, posture–breathing training, etc.) produce greater improvement. [12-15,17-20]. |
|
Type 5: Environment–process-circadian dominant |
Axis 5 |
Light/noise/workflow disruptions → increased nocturnal awakenings and reduced sleep continuity. [1-4]. |
Document environmental exposures and workflow time-points; PSQI/ISI stratification [39,40]. |
Light/noise control and circadian/behavioral rhythm optimization (QI measures) show greater sensitivity [1-4]. |
|
Type 6: Multi-axis overlap / complex COMISA |
Multiple axes |
Two or more axes are overt (e.g., Axis 4 + 2 or Axis 1 + 3); SDB plus insomnia-like overlap is common [39-42]. |
After PSQI/ISI + OSA screening, use the dominant/secondary axis combination as an enrollment stratifier (P3/P4) [39-42]. |
Combined interventions and multidisciplinary pathways; stratifying by overt axis combinations better supports interaction-effect testing [39-42]. |
Note: Type 1-6 are candidate definitions. Classification should follow table 2 (P1-P4) and boundary conditions. When evidence is insufficient, use qualifying language such as “candidate” or “suggestive” [1-4,9-11].
Clinical application and practice mapping
To support bedside use while avoiding over-causal interpretation, this section foregrounds “clinical use-practice mapping-quality improvement” as a bridge from mechanisms to practice [1-4]. In perioperative care, this framework should be used as a tool for stratification and differential framing, not as a direct diagnostic conclusion. We suggest the following stepwise approach: (1) use clinical history and standardized instruments to complete baseline stratification for PSD/insomnia and OSA risk (e.g., PSQI, ISI, and STOP-type tools); (2) assess the major driving domains across the five mechanistic axes (pain/medications/inflammation-immune/environment-process/autonomic factors); (3) when clues such as neck-shoulder discomfort, postural abnormalities, posture-related breathing discomfort, or exposures such as intubation, prone positioning, or head-neck immobilization are present, further evaluate CST-DS triggers; and (4) use the resulting “explicit axis combination” to guide individualized intervention bundles and stratified enrollment for research [1-4,39-42].
Clinical examples: If a patient with postural abnormalities develops difficulty initiating sleep, increased awakenings, and morning neck-shoulder discomfort after intubation-related cervical load or positioning-related fixation, accompanied by nocturnal breathing discomfort, these features are consistent with a candidate Type 4 (CST-DS-dominant) presentation. If sleep fragmentation and neck-shoulder pain/headache occur after prone positioning or head-neck immobilization, Axis 1 amplification should be assessed early, with Type 4 or Type 6 (multi-axis overlap) considered [6-8,12-16].
This phenotyping system can support perioperative risk stratification and coordinated referral (different “active axes” can trigger different specialty/process interventions), and it can facilitate testing of cross-axis interaction effects in stratified trial designs [39-42]. Importantly, this classification remains hypothesis-generating. While associative evidence exists, direct perioperative causal evidence is limited; CST-DS and glymphatic-like pathways in particular require validation [1-4,6-8,28-34].
Discussion
Recent research on sleep, brain metabolite clearance, CSF flow, and the glymphatic-like system suggests that sleep fragmentation, inflammatory responses, and alterations in cerebrovascular/endothelial function may jointly influence intracranial fluid transport and clearance, providing biological plausibility for the “inflammation-immune-CSF/venous axis” (Axis 2) [28-30]. However, translating these mechanisms directly to perioperative PSD requires caution. Direct evidence in perioperative populations remains limited, and future studies should use quantifiable imaging/biomarkers alongside longitudinal sleep measurements to test causal chains and temporal ordering. For conceptual provenance, we cite foundational work on the paravascular pathway, arterial pulsation-driven CSF-ISF exchange, and sleep-facilitated metabolite clearance, along with subsequent reviews to establish the mechanistic lineage [28-32].
In anesthesia/sedation contexts, animal studies suggest that intracranial fluid exchange under anesthesia may couple with electrophysiologic and autonomic markers, offering a mechanistic link for the hypothesis that perioperative exposures could influence clearance pathways. Narrative perioperative syntheses also propose that sleep disturbance, neuroinflammation, and glymphatic-like dysfunction may jointly contribute to PND, but the evidence tier and falsifiable clinical pathways still require reinforcement through human studies [33,34].
The central value of this multi-axis classification is that it places the traditional PSD “risk-domain-symptom-spectrum” vocabulary and emerging constructs such as CSDS/CST-DS into a single structure, allowing observations across disciplines to map onto testable mechanistic hypotheses and study-design entry points [1-5,9,10]. To strengthen the empirical grounding of these concepts and to align with methodological recommendations for narrative integrative reviews that emphasize “testability and verification,” we propose preregisterable predictions and recommend concurrent collection of mechanistic markers [6-8].
Prediction 1 (physiologic falsifiability across types)
Among PSD patients prospectively classified as Type 4 (CST-DS-dominant), a standardized cervical/peripheral intervention (e.g., posture–breathing training and/or a reproducible soft-tissue intervention) should produce a significantly greater 24-72-hour improvement in HRV (e.g., RMSSD or high-frequency power) relative to baseline than in Type 1 (autonomic-endocrine stress-dominant). A phenotype-stratified randomized controlled trial is recommended, with HRV and sleep outcomes (PSQI/ISI or objective sleep metrics) as co-primary endpoints and a prespecified Type × Intervention interaction test [9,10,12-16,39,40].
Prediction 2 (quantifiable mechanistic chain for Axis 2)
If Axis 2 is dominant in certain phenotypes, sleep improvement after intervention should be accompanied by concordant decreases or dynamic shifts in inflammatory/neuroinflammatory markers. A tiered biomarker strategy is feasible: plasma/serum inflammatory markers as lower-burden proxies; and, where ethically permissible, exploratory CSF biomarkers to test coherence of the “inflammation–sleep–neurocognitive outcomes (delirium/PND)” chain [21-25,28-32,34].
Prediction 3 (falsifiability in clinical outcomes)
If the CST-DS trigger axis is a key driver of Type 4, then under high-exposure conditions (intubation-related cervical load, hyperextension/traction, positioning-related fixation), Type 4 patients should show a larger increase in sleep fragmentation, pain-sleep interaction, and delirium risk. Correspondingly, interventions targeting the “cervical load-posture-breathing” chain should yield greater marginal benefit (effect modification) [37,38,12-16,35,21,28].
Limitations
Clinical experience and part of the supporting evidence for this framework largely come from Asian practice settings. Differences in body habitus, comorbidity profiles, and care pathways may influence baseline PSD risk and sensitivity to CST-DS triggers; external validity therefore requires multicenter confirmation. In addition, this is a narrative integrative review rather than a preregistered systematic review, and thus it is subject to topic-selection bias and cannot provide quantitative pooled estimates. We address this by explicitly presenting evidence-strength gradients in Table 1 and by consistently using qualified language to avoid over-causal interpretation [6-8].
Conclusions
Perioperative sleep disturbances arise from the convergence of multiple pathways, including psychological factors, pain, medication exposures, inflammatory-immune responses, and environmental/care-process factors, as well as a potentially under-recognized cervical-posture-ventilation-autonomic trigger chain [1-5,35].
We propose a multi-axis, hypothesis-generating classification framework in which five mechanistic axes jointly define perioperative sleep phenotypes. Through evidence-strength grading and explicit boundary conditions, the framework incorporates the “cervical-posture-ventilation-autonomic” trigger axis into the PSD explanatory system in a structured way [6-6,12-16,21-27]. Its core value is the generation of falsifiable clinical predictions and study-design elements (e.g., stratified randomized trials or prospective cohorts with phenotype-based enrolment, stratification, and interaction-effect testing) that can be implemented and validated in future multicentre research when feasible [6-8].
Part II will translate this framework into bedside-operational screening checklists and stratified intervention pathways and will propose a feasible validation agenda. Perioperative sleep interventions-including both nonpharmacologic and pharmacologic strategies-may reduce delirium risk or improve postoperative neurocognitive outcomes, providing a practical lever for phenotype-stratified sleep management [27].
Author Contributions
QY: Conceptualization, Methodology, Drafting of the initial manuscript;
FFL: Literature synthesis, Drafting of the initial manuscript;
MYC: Figures/Tables and Visualization;
YES/JFW: Critical review and revision of key content;
WC: Conceptualization, Supervision, Funding acquisition, Final approval.
All authors approved the final version and agree to be accountable for all aspects of the work.
Conflicts of Interest
None.
Funding
National Natural Science Foundation of China (Project Approval Number: 81200858); Jiangsu Province 333 High-level Talent Training Project [Certificate No.: (2022) No. 3-10-007].
Clinical Trials from Nanjing Drum Tower Hospital, Affiliated Hospital of Medical School, Nanjing University, the Huai'an Matching Assistance Special Project (2024-2025.
AI Use Statement
- We used large language model-based tools (e.g., ChatGPT) solely for language editing, formatting support, and improving clarity.
- AI tools were not used to generate new scientific conclusions, fabricate evidence, or replace the authors’ scholarly judgment; all key ideas, structure, and final wording were reviewed and revised by the author team, which takes full responsibility for the manuscript.
- The authors assume full responsibility for the accuracy, originality, and citation integrity of all content.
References
- Lin D, Huang X, Sun Y, Wei C, Wu A (2021) Perioperative sleep disorder: A review. Frontiers in Medicine 8.
- Sipilä RM, Kalso EA (2021) Sleep well and recover faster with less pain-A narrative review on sleep in the perioperative period. J Clin Med 10: 2000.
- Butris N, Tang E, Pivetta B, He D, Saripella A, et al. (2023) The prevalence and risk factors of sleep disturbances in surgical patients: A systematic review and meta-analysis. Sleep Med Rev 69.
- Nelson MJ, Yu DA, Ha AVH, Wakefield MR, Fang Y (2025) Causes and effects of postoperative sleep disorders and treatment strategies for preoperative, intraoperative, and postoperative Settings-A narrative review. Clocks Sleep 7: 29.
- Finan PH, Goodin BR, Smith MT (2013) The association of sleep and pain: An update and a path forward. J Pain 14: 1539-1552.
- Green BN, Johnson CD, Adams A (2006) Writing narrative literature reviews for peer-reviewed journals: secrets of the trade. J Chiropr Med 5: 101-117.
- Ferrari R (2015) Writing narrative style literature reviews. Medical Writing 24: 230-235.
- Sukhera J (2022) Narrative reviews: Flexible, rigorous, and practical. J Grad Med Educ 14: 414-417.
- Yin Q, Cheng MY, Wang S, Sun YE, Wang JF, et al. (2025) Cervicogenic sleep disorder syndrome: Definition and classification recommendations. Transl Perioper Pain Med 12: 746-767.
- Yin Q, Zheng Y, Wang S, Sun YE, Wang JF, et al. (2025) “Postural alignment first, symptom tailored” integrated pathway as a multimodal peripheral intervention strategy for pain and cervical disease (Part 1). Transl Perioper Pain Med 12: 716-733.
- Chaibi A, Russell MB (2012) Manual therapies for cervicogenic headache: A systematic review. J Headache Pain 13: 351-359.
- Leung JM, Sands LP, Newman S, Meckler G, Xie Y, et al. (2015) Preoperative Sleep Disruption and Postoperative Delirium. J Clin Sleep Med 11: 907-913.
- Leung JM, Tang C, Do Q, Sands LP, Tran D, et al. (2023) Sleep loss the night before surgery and incidence of postoperative delirium in adults 65–95 years of age. Sleep Med 105: 61-67.
- Badau A, Badau D, Cengiz SS, Coşkun ES (2024) The relationship between sleep quality and posture in young adults. Life 14: 1244.
- Yabe Y, Hagiwara Y, Sekiguchi T, Sugawara Y, Tsuchiya M, et al. (2022) Sleep disturbance is associated with neck pain: a 3-year longitudinal study. BMC Musculoskelet Disord 23: 459.
- Lobbezoo F, Visscher CM, Naeije M (2004) Impaired health status, sleep disorders and pain in the craniomandibular and cervical spinal regions. Eur J Pain 8: 23-30.
- Hadamus A, Wojda A, Białoszewski D (2021) Can the sleep quality of patients with chronic neck pain be improved by muscle energy techniques combined with Swedish massage? Complement Ther Clin Pract 44.
- Lee WH, Ko MS (2017) Effect of sleep posture on neck muscle activity. J Phys Ther Sci 29: 1021-1024.
- Jeon MY, Jeong HC, Lee SW, Choi W, Park JH, et al. (2014) Effects of a suitable pillow on cervical lordosis and subjective sleep quality. Tohoku J Exp Med 233: 183-188.
- Vanti C, Banchelli F, Marino C, Puccetti A, Guccione AA, et al. (2019) Effectiveness of a “spring pillow” versus education in chronic nonspecific neck pain: A randomized controlled trial. Phys Ther 99: 1177-1188.
- Pang JC-Y, Tsang SM-H, Fu AC-L (2021) The effects of pillow designs on neck pain, waking symptoms, neck disability, sleep quality and spinal alignment in adults: A systematic review and meta-analysis. Clin Biomech (Bristol) 85:105353. doi:10.1016/j.clinbiomech.2021.105353.
- Lavin RA, Pappagallo M, Kuhlemeier KV (1997) Cervical pain: a comparison of three pillows. Arch Phys Med Rehabil 78(2):193-198. doi:10.1016/S0003-9993(97)90263-X.
- Haack M, Simpson N, Sethna N, Kaur S, Mullington J (2020) Sleep deficiency and chronic pain: Potential underlying mechanisms and clinical implications. Neuropsychopharmacology 45: 205-216.
- Bjurström MF, Irwin MR, Chen DC, Smith MT, Montgomery A (2021) Sex differences, sleep disturbance and risk of persistent pain after groin hernia surgery. J Pain 22: 1360-1370.
- Buysse DJ, Reynolds CF, Monk TH, Berman SR, Kupfer DJ (1989) The pittsburgh sleep quality index: A new instrument for psychiatric practice and research. Psychiatry Res 28: 193-213.
- Bastien CH, Vallières A, Morin CM (2001) Validation of the insomnia severity index as an outcome measure for insomnia research. Sleep Med 2: 297-307.
- Chung F, Yegneswaran B, Liao P, Chung SA, Vairavanathan S, et al. (2008) STOP questionnaire: A tool to screen patients for obstructive sleep apnea. Anesthesiology 108: 812-821.
- Thomas TL, Rampam S, Nithagon P, Goh GS (2024) Increased risk of postoperative complications in patients who have obstructive sleep apnea undergoing total joint arthroplasty: A systematic review and meta-analysis. J Arthroplasty 39: 3119-3127.e1.
- Vacas S, Kurien P, Maze M (2013) Sleep and anesthesia - Common mechanisms of action. Sleep Med Clin 8: 1-9.
- Cheng C, Wang H, Cong P, Huang X, Wu T, et al. (2022) Targeting neuroinflammation as a preventive and therapeutic approach for perioperative neurocognitive disorders. J Neuroinflammation 19: 297.
- Xiao MZ, Liu CX, Zhou LG, Yang Y, Wang Y (2023) Postoperative delirium, neuroinflammation, and influencing factors of postoperative delirium: A review. Medicine 102: e32991.
- Guo H, Li LH, Lv XH, Su FZ, Chen J, et al. (2024) Association between preoperative sleep disturbance and postoperative delirium in elderly: A retrospective cohort study. Nat Sci Sleep 16: 389-400.
- Taylor J, Parker M, Casey CP, Tanabe S, Kunkel D, et al. (2022) Postoperative delirium and changes in the blood-brain barrier, neuroinflammation, and cerebrospinal fluid lactate: A prospective cohort study. Br J Anaesth 129: 219-230.
- Lu Y, Li YW, Wang L, Lydic R, Baghdoyan HA, et al. (2019) Promoting sleep and circadian health may prevent postoperative delirium: A systematic review and meta-analysis of randomized clinical trials. Sleep Med Rev 48.
- Tang X, Li J, Li Y (2023) The effect of sleep interventions on postoperative delirium: a systematic review and meta-analysis. Perioper Med 12: 34.
- Iliff JJ, Wang M, Liao Y, Plogg BA, Peng W, et al. (2012) A paravascular pathway facilitates CSF flow through the brain parenchyma and the clearance of interstitial solutes, including amyloid β. Sci Transl Med 4: 147ra111.
- Iliff JJ, Wang M, Zeppenfeld DM, Venkataraman A, Plog BA, et al. (2013) Cerebral arterial pulsation drives paravascular CSF-interstitial fluid exchange in the murine brain. J Neurosci 33: 18190-18199.
- Xie L, Kang H, Xu Q, Chen MJ, Liao Y, et al. (2013) Sleep drives metabolite clearance from the adult brain. Science 342: 373-377.
- Rasmussen MK, Mestre H, Nedergaard M (2018) The glymphatic pathway in neurological disorders. Lancet Neurol 17: 1016-1024.
- Rasmussen MK, Mestre H, Nedergaard M (2022) Fluid transport in the brain. Physiol Rev 102: 1025-1151.
- Hablitz LM, Vinitsky HS, Sun Q, Stæger FF, Sigurdsson B, et al. (2019) Increased glymphatic influx is correlated with high EEG delta power and low heart rate in mice under anesthesia. Sci Adv 5: eaav5447.
- Dong R, Liu W, Han Y, Wang Z, Jiang L, et al. (2024) Influencing factors of glymphatic system during perioperative period. Front Neurosci 18.
Table of Contents
- Abstract
- Keywords
- Introduction
- From Five Mechanistic Axes to Actionable Phenotypes: Evidence Gradients, Workflow-Based Derivation, and Candidate Types (Type 1-6)
- Discussion
- Conclusions
- Author Contributions
- Conflicts of Interest
- Funding
- Figure 1
- Figure 2
- Figure 3
- Figure 4
- Figure 5
- Table
- Table
- Table
- References