Translational Perioperative and Pain Medicine (ISSN: 2330-4871)
ARTICLE DOI: 10.31480/2330-4871/160
Perspective | Volume 9 | Issue 3 Open Access
Therapeutic Considerations in Chronic Serotonin Syndrome: A Case Report
Uday Agrawal, MD1#, Yihang Qi, MD1,2# and Jingping Wang, MD, PhD1*
1Department of Anesthesia, Critical Care and Pain Medicine, Massachusetts General Hospital, Harvard Medical School, Boston, Massachusetts, USA
2Department of Breast Surgical Oncology, National Cancer Center/National Clinical Research Center for Cancer/Cancer Hospital, Chinese Academy of Medical Sciences and Peking Union Medical College, Beijing, 100021, China
#Contributed equally as the first author
Jingping Wang, MD, PhD, Department of Anesthesia Critical Care and Pain Medicine, Massachusetts General Hospital, Harvard Medical School, 55 Fruit Street (GRJ-420), Boston, MA, USA, Tel: 617-643-2729, E-mail: jwang23@mgh.harvard.eduEditor: Yuan-Xiang Tao, Ph.D., M.D., Professor and Vice Chair, Director, Center for Pain Medicine Research, Department of Anesthesiology, Editor in Chief, The Translational Perioperative and Pain Medicine, Rutgers, The State University of New Jersey, New Jersey Medical School, 185 S. Orange Ave., MSB, E-661, Newark, NJ 07103, USA, Tel: 973-972-9812; E-mail: yt211@njms.rutgers.edu
Received: June 22, 2022 | Accepted: August 20, 2022 | Published: September 12, 2022
Citation: Agrawal U, Qi Y, Wang J. Therapeutic Considerations in Chronic Serotonin Syndrome: A Case Report. Transl Perioper & Pain Med 2022; 9(2):464-469
Abstract
An elderly patient with depression and chronic back pain presents with several months of new onset agitation, insomnia, hyperhidrosis, hypertension, tachycardia, nausea, and vomiting. Her pain quality is unchanged, and basic labs and imaging including cardiac echocardiography are normal. Notably, her medication regimen includes opioids and anti-depressants raising concern for chronic serotonin syndrome. Here, we review the diagnosis of serotonin syndrome and therapeutic considerations for management of the challenging condition.
Case Presentation
An 81-year-old woman with depression and long standing chronic back pain presents with several months of new onset agitation, excessive sweating at night associated with insomnia, nausea, vomiting, poor appetite, dizziness with multiple falls, elevated blood pressures, and an increased heart rate.
Her back pain is secondary to spinal stenosis, facet arthropathy, sacroiliac joint syndrome, degenerative disc disease, and failed back surgery syndrome. She reports no change in her baseline quality and severity of pain over the past several months and her mood has remained stable. The pain is exacerbated by sitting (spinal flexion) and improved by standing (spinal extension) and she denies any progressive weakness, numbness, or functional bowel or bladder symptoms. Her physical exam is notable for bilateral tenderness to palpation over the sacroiliac joints, lumbar paraspinal muscles, and lumbar facet and spinous processes bilaterally, which has remained similar over the past several years. She is noted to have intention tremor and hyperreflexia, but no clonus. She had an extensive laboratory workup including a basic metabolic panel (BMP), complete blood count (CBC), thyroid stimulating hormone (TSH), vitamin D, hemoglobin A1c (Hb A1c), c-reactive protein (CRP), erythrocyte sedimentation rate (ESR), anti-nuclear antibody (ANA), rheumatoid factor (RF), double stranded-DNA (dsDNA), anti-Sjogren's syndrome A (SS-A, Ro), anti-Sjogren's syndrome B (SS-B, La), cyclic citrullinated peptide (CCP), and human leukocyte antigen B27 (HLA-B27), all of which were normal. She also had computed tomography (CT) scans of her head, chest, and abdomen in addition to a transthoracic cardiac echocardiogram (TTE) which were normal.
In the past she has trialed numerous interventions including multiple epidural steroid injections, a facet joint injection, a spinal cord stimulator, bilateral lumbar medial branch blocks, and Synvisc injections of her left knee which have provided some pain relief but have become less effective over time. She has also undergone four prior back surgeries including L2 through L5 fusion with transpedicular screws and vertical joining rods, discectomy and interbody fusion at L3-4 and L4-5, and posterior decompression which have not brought significant relief of her pain.
For the past several years, her pain has been managed intermittently with opioids (including buprenorphine, methadone, and oxycodone) and anti-depressants (including duloxetine, lamotrigine, and mirtazapine). Tapers of her opioids have been attempted and resulted in opioid withdrawal and poor pain control leading to exacerbation of her depression. Strategies to taper her anti-depressants or trial alternative agents have similarly resulted in severe depression and worsening of her chronic pain. Of note, methadone 5 mg q12h has been added to her pain regimen over the past six months. Her other current medication regimen consists of oxycodone 5 mg q12H prn, pregabalin 100 mg daily, acetaminophen 1g q8h, celecoxib 100 mg q12h, duloxetine 40 mg daily, lamotrigine 100 mg daily, mirtazapine 7.5 mg nightly, metoprolol ER 50 mg daily, irbesartan 75 mg daily, amlodipine 5 mg daily, atorvastatin 10 mg daily, omeprazole 20 mg q12h, melatonin 6 mg daily, coenzyme Q10 200 mg daily, and vit D3 2000u daily.
Given the constellation of new symptoms in this patient (agitation, insomnia, hyperhidrosis, nausea, vomiting, dizziness, hypertension, tachycardia, tremor, hyperreflexia), unrevealing laboratory and imaging workup, and polypharmacy for management of her comorbid depression and chronic pain, a multidisciplinary conference with pain specialists, psychiatrists, geriatricians, and neurologists was held and the diagnosis of chronic serotonin syndrome was considered.
The patient's duloxetine was tapered and ultimately discontinued within four weeks. Her methadone and pregabalin were also discontinued within four weeks as well. The mirtazapine dose was slowly increased from 7.5 mg nightly to 45 mg nightly, with consideration for quetiapine 25 mg nightly or CBD oil if insomnia were to persist. Her pain has been controlled well with acetaminophen 1g q8h, celecoxib 100 mg q12h, and a TENS unit daily. Her symptoms of agitation, tremor, hyperreflexia, nausea, vomiting, hypertension and tachycardia were significantly improved.
Discussion
Serotonin syndrome -also called serotonin toxicity- is classically characterized as a triad of cognitive, autonomic, and neuromuscular symptoms induced by a state of elevated serotonin levels [1-7]. The symptoms range in severity from mild to lethal and may be dependent on serotonin concentration and action at specific receptor sites [3]. Cognitive symptoms include anxiety, insomnia, agitation, delirium or confusion. Autonomic symptoms include diaphoresis, tachycardia, tachypnea, hypertension, diarrhea, nausea, vomiting, and hyperthermia. Neuromuscular findings include tremor, shivering, rigidity, hyperreflexia, myoclonus, and clonus [1,3-5] (see Figure 1).
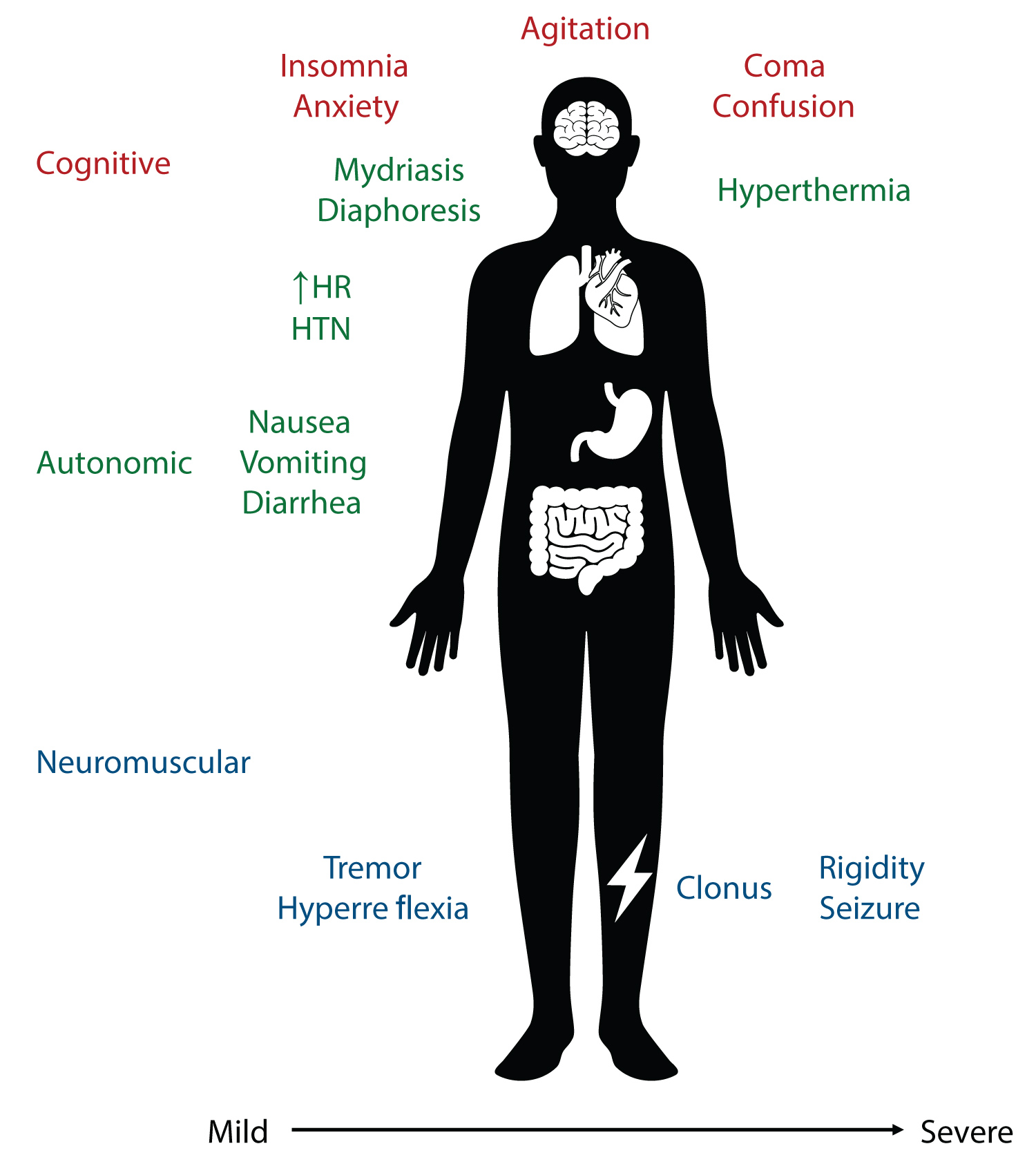
Figure 1:
Figure 1: Common symptoms of serotonin syndrome.
The symptoms of serotonin syndrome are traditionally described as either cognitive, autonomic, or neuromuscular and they range in severity from mild to moderate and severe. The cognitive symptoms include insomnia, anxiety (mild), agitation (moderate), and coma or confusion (severe). Autonomic symptoms include nausea, vomiting, diarrhea (mild), mydriasis, diaphoresis, hypertension, tachycardia (moderate), and hyperthermia (severe). Neuromuscular symptoms include tremor, hyperreflexia (mild), clonus (moderate), or rigidity and seizure (severe).
Notably, serotonin syndrome is a clinical diagnosis and no specific laboratory or imaging findings are sensitive or specific for the condition [4], though they may be helpful in exonerating alternative diagnoses. The differential diagnosis of a suspected serotonin syndrome is broad and includes neuroleptic malignant syndrome, anticholinergic syndrome, and medication or substance withdrawal states in addition to primary metabolic, endocrine, or infectious etiologies [7].
Given these diagnostic challenges, four criteria have been proposed as examined in a recent review by Chiew and Buckley [7] which categorizes the symptoms of serotonin syndrome in effort to standardize diagnosis. Of these criteria (Serotonin syndrome score [8], Sternbach [9], Radomski [10], and Hunter [11]), the Sternbach, Radomski, and Hunter are more widely used and Hunter is considered the gold standard. However, the authors note major shortcomings of each of these criteria and propose their own criteria more informed by the clinical scenario and plausibility of a serotonin toxidrome based on the patient's specific drug history in addition to the manifested symptoms.
In particular, there are several drugs that increase the concentration of or amplify the effects of serotonin and predispose a patient to serotonin syndrome [2-7,12,13]. Of relevance to the presented case, the phenylpiperidine opioids (including methadone) and many anti-depressants (including duloxetine) [2,3,7,12,13] act as serotonin reuptake inhibitors and have been implicated in serotonin syndrome. Methadone has been found to lead to serotonin syndrome even after one dose in a patient on duloxetine and oxycodone [13]. Numerous drugs including opioids are also linked to serotonin syndrome though the evidence is not as robust [7,12]. For example, oxycodone has been associated with serotonin syndrome possibly secondary to drug-drug interactions in a few case reports [14-16]. Similarly, lamotrigine in high concentrations in vitro has been shown to inhibit uptake of serotonin in mice [17] and it has been associated with serotonin syndrome in a case report [18].
The patient presented here has undergone titration of medications associated with serotonin syndrome (methadone and duloxetine) over several months and has undergone an extensive workup to determine the cause of her symptoms. She manifested cognitive symptoms of agitation, restlessness, and insomnia, autonomic symptoms of tachycardia, hypertension, diaphoresis, nausea, vomiting, and dizziness with multiple falls, and neuromuscular symptoms including tremor and hyperreflexia. With this presentation, she satisfies all of the diagnostic criteria for serotonin syndrome [7], though experienced these symptoms over a time scale of several months, which presented chronically (Table 1).
Table 1: Hunter Criteria: The most widely used criteria to diagnose serotonin syndrome.
Hunter Criteria
In the presence of a serotonergic agent, any of the following:
|
Spontaneous clonus |
|
Inducible clonus and agitation or diaphoresis |
|
Ocular clonus and agitation or diaphoresis |
|
Tremor and hyperreflexia |
|
Hypertonic and temperature > 38 °C and ocular clonus or inducible clonus |
Typically symptoms of serotonin syndrome have been reported to occur within one hour of a precipitating event in about 30% of patients and within six hours in 60% of patients [7,19]. However, more recently serotonin syndrome has also been observed on a longer, more chronic time scale [20-24]. Prakash, et al. performed a retrospective study and found several cases of chronic serotonin syndrome with symptoms persisting on a time scale of 6-24 weeks with no clear alternative diagnoses and resolution of symptoms with cyproheptadine [20]. They hypothesize that mildly elevated serotonin levels over a longer time period can lead to sustained upper motor neuron signs and associated symptoms resulting in a chronic form of serotonin syndrome [20]. Several case reports of serotonin syndrome have similarly been observed on this longer time scale - for example, Lamberg, et al. noted a patient treated with additional escalating doses of serotonergic agents developed tremors, akathisia, a dry mouth, and inducible clonus consistent with a diagnosis of serotonin syndrome over the course of two months and had resolution of symptoms after discontinuing the serotonergic agents [22]. As in the case presented here, these reports demonstrate that while serotonin syndrome is often an acute emergency, more indolent prolonged time courses of serotonin syndrome may also occur and recognition of this condition is critical.
Once serotonin syndrome is recognized, management consists of discontinuing the causal agents, considering use 5-HT2A antagonists, and supportive care of the associated symptoms often with use of benzodiazepines [4,5] (see Figure 2). The intensity of the supportive care is determined by the severity of a patient's symptoms and may range from symptomatic support with benzodiazepines to possible intensive care unit admission for neuromuscular blockade and intubation in the setting of severe hyperthermia. In general the prognosis of serotonin syndrome is favorable, though it is dependent on the severity of the initial symptoms, the time frame of recognition, and appropriate treatment of the complications. Mild to moderate cases often resolve within 24 hours of cessation of the causal agent and supportive care, though the symptoms may persist for longer depending in the half-life of the associated drugs [4,25]. Lethal cases of serotonin syndrome have also been reported, often secondary to co-ingestion of multiple substances (especially monoamine oxidase inhibitors) and less likely following selective serotonin reuptake inhibitor overdose alone [25].
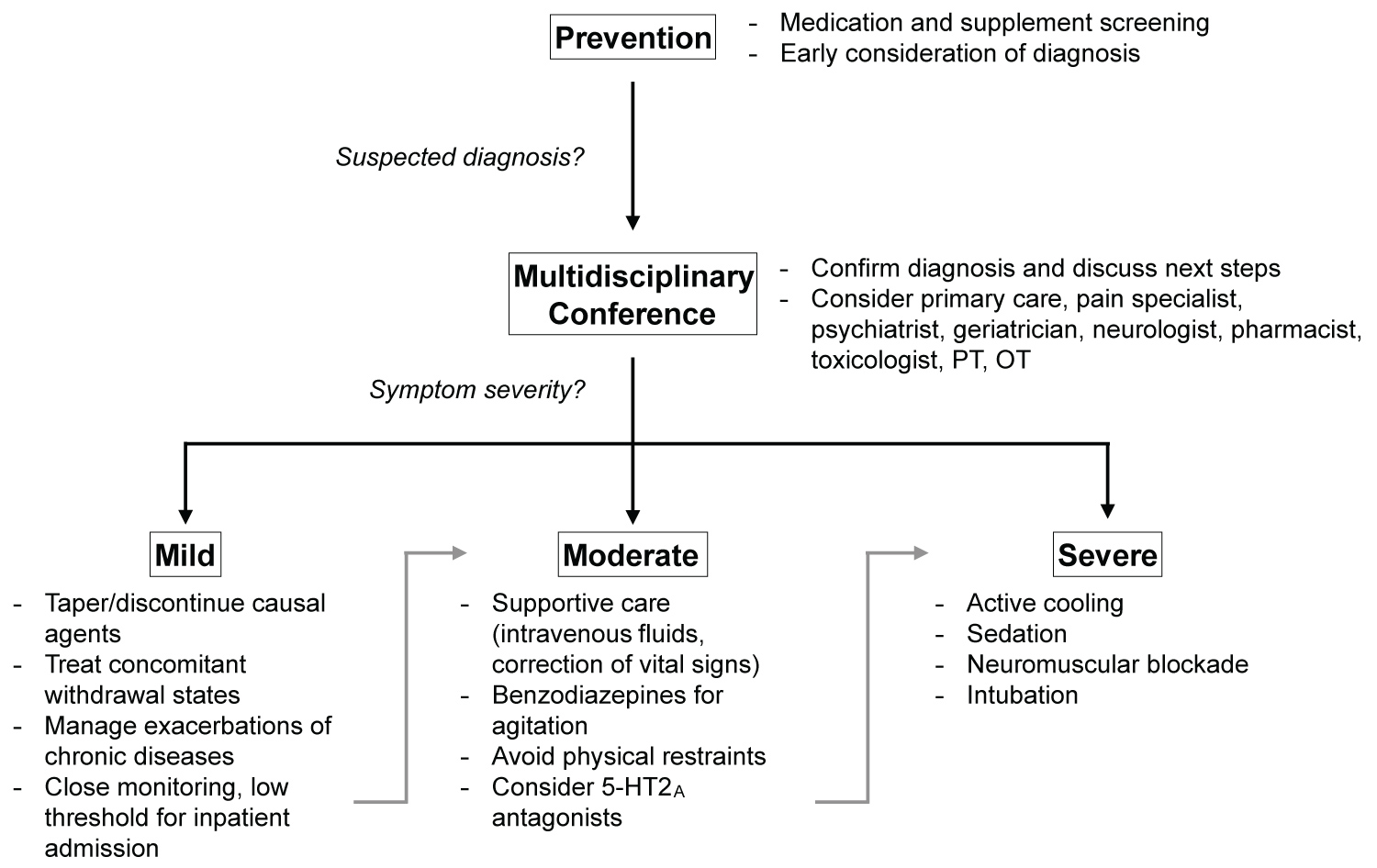
Figure 2:
Figure 2: General approach to treatment of serotonin syndrome.
The primary approach to treating serotonin syndrome is prevention. This is especially critical in elderly populations with polypharmacy. Once the diagnosis is suspected, it should be confirmed with a multidisciplinary discussion which can also help to determine the optimal therapeutic interventions. The treatment plan can then be implemented in the appropriate setting with contingencies to treat the comorbid conditions and escalate care as necessary.
In the case presented here the decision of which medications to discontinue and strategies to taper is more nuanced as depression and chronic pain are strongly associated [26,27] and polypharmacy in elderly patients is known to have an overall adverse effect [28]. As a result, efforts to reduce serotonergic opioids may lead not only to symptoms of opioid withdrawal, but also worsening of chronic pain and an associated exacerbation of depression. Similarly, tapering of anti-depressants can worsen mood and as a result chronic pain, and managing these symptoms with alternative therapies further contributes to polypharmacy.
It is critical to consider a multidisciplinary approach to treatment of polypharmacy induced serotonin syndrome, especially in elderly populations with depression and chronic pain. Initial prevention of the syndrome remains the mainstay of management and can be facilitated by thorough medication and supplement screenings in patients at risk. Once the diagnosis is suspected, a discussion with multiple specialists may be helpful not only to confirm the diagnosis, but also to consider the most appropriate next steps including the setting of treatment. This decision is often made based on the severity of symptoms of the serotonin syndrome (eg. hyperthermia, autonomic fluctuations), but additional emphasis should also be placed on the risk of exacerbating underlying symptoms of depression or pain as these may place the patient at risk for acute exacerbations requiring inpatient monitoring and treatment. For example, in the case presented the patient has experienced suicidal ideation in the past and abrupt discontinuation of her anti-depressants would likely pose greater risk than tapering of her opioids and may warrant inpatient admission despite her relatively mild symptoms. A multidisciplinary conference can therefore help to establish which medications to discontinue at which time, especially if multiple medications may be contributing. Depending on the severity of the serotonin syndrome, a taper of serotonergic medications may also be possible to avoid withdrawal states if the patient can be monitored closely. Finally, alternative treatments must be considered if necessary that carry a lower risk of precipitating serotonin syndrome but are still effective therapies.
In this patient the decision was made to monitor her closely in the outpatient setting with a very low threshold for inpatient admission. Her mirtazapine (which manifests some serotonin antagonism) was increased to counteract the possible depressed mood prior to tapering of her duloxetine and her mood remained stable. Non-pharmaceutical pain strategies including a TENS unit and physical therapy along with her previously prescribed breakthrough oxycodone were successful in preventing an exacerbation of her pain while discontinuing the methadone. Additional strategies including buprenorphine - which may have an anti-depressant effect [29-31] - were considered, but unfortunately it was trialed unsuccessfully in the past. There was also a discussion regarding the emerging data about the impact of ketamine on both pain and depression, though additional trials are necessary [32,33] and it was not used for this patient. Overall, her symptoms have significantly improved with this approach.
Conclusion
The assessment of serotonin syndrome is both challenging and necessary, especially in elderly patients with depression and chronic pain who are at risk for the condition. While serotonin syndrome is typically thought to be an acute event, several cases have also been reported with symptoms persisting over the course of weeks to months and it is important to consider the diagnosis in patients with polypharmacy. Once it is diagnosed, a multidisciplinary approach to management may be helpful to ensure optimal treatment of both serotonin syndrome and the patient's comorbid conditions.
Declaration of Interests
The authors have no conflicts of interest to declare.
Author Contributions
Uday Agrawal: This author contributed to acquisition of data, writing the manuscript, interpretation, discussion and revising the final manuscript; Yihang Qi: This author contributed substantially to acquisition of data, writing the manuscript, interpretation, discussion, and revising the final manuscript; Jingping Wang: This author helped with the conception and design, interpretation, writing the draft, revising the final manuscript, and approved of the final submitted manuscript.
Availability of Data and Material
The data generated or analyzed during this study are included in this article, or if absent are available from the corresponding author upon reasonable request.
Acknowledgments
We would like to acknowledge everyone who participated in the multidisciplinary discussion: Dr. Renyu Liu (Anesthesiologist), Dr. James Jia (Hospitalist), Dr. Danzhao Wang (Psychiatry Specialist), Dr. Wenchun Qu (Pain Medicine Specialist), Dr. Jianhua Zhu (Neurologist), Dr. Fang Guo (Internal Medicine), Dr. Peijun Chen (Geriatric Psychiatry), Dr. Cecilia Wang (Psychiatry Specialist), Dr. Julun Hong, Dr. Ze Hui.
References
- Houchens N. Serotonin Syndrome: Preventing, Recognizing, and Treating It. Cleve Clin J Med. 2016;83:810-817. doi:10.3949/ccjm.83a.15129
- Scotton WJ, Hill LJ, Williams AC, Barnes NM. Serotonin Syndrome: Pathophysiology, Clinical Features, Management, and Potential Future Directions. Int J Tryptophan Res IJTR. 2019;12:1178646919873925. doi:10.1177/1178646919873925
- Sun-Edelstein C, Tepper SJ, Shapiro RE. Drug-induced serotonin syndrome: a review. Expert Opin Drug Saf. 2008;7(5):587-596. doi:10.1517/14740338.7.5.587
- Boyer EW, Shannon M. The Serotonin Syndrome. N Engl J Med. 2005;352(11):1112-1120. doi:10.1056/NEJMra041867
- Gillman PK. The serotonin syndrome and its treatment. J Psychopharmacol (Oxf). 1999;13(1):100-109. doi:10.1177/026988119901300111
- Birmes P, Coppin D, Schmitt L, Lauque D. Serotonin syndrome: a brief review. CMAJ. 2003;168(11):1439-1442.
- Chiew AL, Buckley NA. The serotonin toxidrome: Shortfalls of current diagnostic criteria for related syndromes. Clin Toxicol Phila Pa. 2022;60(2):143-158. doi:10.1080/15563650.2021.1993242
- Hegerl U, Bottlender R, Gallinat J, Kuss HJ, Ackenheil M, Moller HJ. The serotonin syndrome scale: first results on validity. Eur Arch Psychiatry Clin Neurosci. 1998;248(2):96-103. doi:10.1007/s004060050024
- Sternbach H. The serotonin syndrome. Am J Psychiatry. 1991;148(6):705-713. doi:10.1176/ajp.148.6.705
- Radomski JW, Dursun SM, Reveley MA, Kutcher SP. An exploratory approach to the serotonin syndrome: an update of clinical phenomenology and revised diagnostic criteria. Med Hypotheses. 2000;55(3):218-224. doi:10.1054/mehy.2000.1047
- Dunkley EJC, Isbister GK, Sibbritt D, Dawson AH, Whyte IM. The Hunter Serotonin Toxicity Criteria: simple and accurate diagnostic decision rules for serotonin toxicity. QJM. 2003;96(9):635-642. doi:10.1093/qjmed/hcg109
- Rastogi R, Swarm RA, Patel TA, Riou B. Case Scenario: Opioid Association with Serotonin Syndrome: Implications to the Practitioners. Anesthesiology. 2011;115(6):1291-1298. doi:10.1097/ALN.0b013e31823940c0
- Kumai M, Maeda Y, Miura M, et al. Serotonin Syndrome Developing Immediately after the Initiation of Low-Dose Methadone Therapy: A Case Report. Case Rep Oncol. 2020;13(1):281-284. doi:10.1159/000506443
- Rosebraugh CJ, Flockhart DA, Yasuda SU, Woosley RL. Visual Hallucination and Tremor Induced by Sertraline and Oxycodone in a Bone Marrow Transplant Patient. J Clin Pharmacol. 2001;41(2):224-227. doi:10.1177/00912700122009926
- Serotonin Syndrome Induced by Fluvoxamine and Oxycodone - Harindra Karunatilake, Nicholas A Buckley, 2006. Accessed May 21, 2022. https://journals.sagepub.com/doi/abs/10.1345/aph.1e671
- Baldo BA. Opioid analgesic drugs and serotonin toxicity (syndrome): mechanisms, animal models, and links to clinical effects. Arch Toxicol. 2018;92(8):2457-2473. doi:10.1007/s00204-018-2244-6
- Southam E, Kirkby D, Higgins GA, Hagan RM. Lamotrigine inhibits monoamine uptake in vitro and modulates 5-hydroxytryptamine uptake in rats. Eur J Pharmacol. 1998;358(1):19-24. doi:10.1016/s0014-2999(98)00580-9
- Kotwal A, Cutrona SL. Serotonin Syndrome in the Setting of Lamotrigine, Aripiprazole, and Cocaine Use. Case Rep Med. 2015;2015:e769531. doi:10.1155/2015/769531
- Mason PJ, Morris VA, Balcezak TJ. Serotonin Syndrome Presentation of 2 Cases and Review of the Literature. Medicine (Baltimore). 2000;79(4):201-209.
- Prakash S, Rathore C, Rana K, Roychowdhury D, Lodha D. Chronic serotonin syndrome: A retrospective study. World J Psychiatry. 2021;11(4):124-132. doi:10.5498/wjp.v11.i4.124
- Alnwick GM. Misdiagnosis of serotonin syndrome as fibromyalgia and the role of physical therapists. Phys Ther. 2008;88(6):757-765. doi:10.2522/ptj.20060208
- Lamberg JJ, Gordin VN. Serotonin Syndrome in a Patient with Chronic Pain Polypharmacy. Pain Med. 2014;15(8):1429-1431. doi:10.1111/j.1526-4637.2012.01468.x
- Prakash S, Rathore C. Cyproheptadine-dependent chronic serotonin syndrome. Neurol India. 2016;64(6):1319-1321. doi:10.4103/0028-3886.193796
- Chechani V. Serotonin syndrome presenting as hypotonic coma and apnea: potentially fatal complications of selective serotonin receptor inhibitor therapy. Crit Care Med. 2002;30(2):473-476. doi:10.1097/00003246-200202000-00033
- Simon LV, Keenaghan M. Serotonin Syndrome. In: StatPearls. StatPearls Publishing; 2022. Accessed June 5, 2022. http://www.ncbi.nlm.nih.gov/books/NBK482377/
- Haythornthwaite JA, Sieber WJ, Kerns RD. Depression and the chronic pain experience. Pain. 1991;46(2):177-184. doi:10.1016/0304-3959(91)90073-7
- Fishbain DA, Cutler R, Rosomoff HL, Rosomoff RS. Chronic Pain-Associated Depression: Antecedent or Consequence of Chronic Pain? A Review. Clin J Pain. 1997;13(2):116-137.
- Hajjar ER, Cafiero AC, Hanlon JT. Polypharmacy in elderly patients. Am J Geriatr Pharmacother. 2007;5(4):345-351. doi:10.1016/j.amjopharm.2007.12.002
- Serafini G, Adavastro G, Canepa G, et al. The Efficacy of Buprenorphine in Major Depression, Treatment-Resistant Depression and Suicidal Behavior: A Systematic Review. Int J Mol Sci. 2018;19(8):2410. doi:10.3390/ijms19082410
- Falcon E, Browne CA, Leon RM, et al. Antidepressant-like Effects of Buprenorphine are Mediated by Kappa Opioid Receptors. Neuropsychopharmacology. 2016;41(9):2344-2351. doi:10.1038/npp.2016.38
- Namchuk AB, Lucki I, Browne CA. Buprenorphine as a Treatment for Major Depression and Opioid Use Disorder. Adv Drug Alcohol Res. 2022;0. doi:10.3389/adar.2022.10254
- Schoevers RA, Chaves TV, Balukova SM, Rot M aan H, Kortekaas R. Oral ketamine for the treatment of pain and treatment-resistant depression. Br J Psychiatry. 2016;208(2):108-113. doi:10.1192/bjp.bp.115.165498
- Kurdi MS, Theerth KA, Deva RS. Ketamine: Current applications in anesthesia, pain, and critical care. Anesth Essays Res. 2014;8(3):283-290. doi:10.4103/0259-1162.143110